Blackbird / Warbler Group
The blackbird/warbler group may have Caribbean origins. The Phaenicophilidae are the basal group. Seven species of Phaenicophilidae are confined to Hispaniola, two to Puerto Rico, and one to Jamaica. The other species, the Western Spindalis, occurs in the Bahamas, Cuba and associated islands, and Cozumel. (There is also one breeding record on the mainland, from south Florida in 2009.) Finally, the Wrenthrush lives in Costa Rica and Panama. The two Cuban Teretistris warblers may also belong in Phaenicophilidae.
It's unknown whether these species represent the geographical origins of the clade, or are relict populations of an otherwise more widespread group of extinct passerines. The combined effects of sea level rise and fall and the process that included the consolidation of the Costa Rica/Panama island arc into the Isthmus of Panama could have played an important role in the origins of these families.
Incertae Sedis: Teretistris
1 genera, 2 species
Klicka et al. (2007) and Weir et al. (2009) placed Spindalis sister to the Cuban Teretistris. However, Barker et al. (2013) only found a connection in the cytochrome-b tree. Their overall results placed Teretistris in a clade with Parulidae, Icteridae, Icteria and Zeledonia. However, it is not clear whether Teretistris is sister to Icteridae, Phaenicophilidae, Parulidae, or some combination thereof. As a result, I list them Incertae sedis, before Phaenicophilidae, Icteridae, and Parulide. There's still a lot of uncertainty here.
- Yellow-headed Warbler, Teretistris fernandinae
- Oriente Warbler, Teretistris fornsi
Phaenicophilidae: Palm-Tanager and allies P.L. Sclater, 1886
6 genera, 11 species
Previously, Nesospingus, Calyptophilus, Phaenicophilus, and Spindalis were in the tanager family and Microligea and Xenoligea were in the warbler family. This group (minus Spindalis) was treated as a subfamily of Parulidae in earlier versions of the TiF checklist based on a combination of Burns (1997), Yuri and Mindell (2002), Burns et al. (2003), Klein et al. (2004), Klicka et al. (2007), and Weir et al. (2009), and some guesswork. The more extensive analysis of Barker et al. (2013) finds that these taxa don't seem to belong to either Parulidae or Icteridae, and helped clarify which taxa belong here. The current arrangement is due to Oliveros et al. (2019).
 |
 |
| Western Spindalis | Phaenicophilidae tree |
|---|
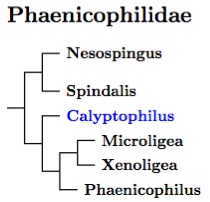
The phylogeny is Oliveros et al. (2019), with help from Barker et al. (2015) for Xenoligea. There is a good chance that Teretistris belongs in here too, but the evidence is weaker for that.
Oliveros et al. (2019) consider the two Hispaniolan Chat-Tanagers to be basal in Phaenicophilidae, followed by the Wrenthrush (Zeledonia), which can be found in Costa Rica and Panama. One of the other two groups is also Hispaniolan, and consists of the Microligea and Xenoligea warblers and the Phaenicophilus Palm-Tanagers. All three genera are sister to the Puerto Rican Tanager (Nesospingus). As it looks somewhat like a female Spindalis, its not surprising the four Spindalis species are its nearest relatives. They are present in the Bahamas and all four major island of the Greater Antilles.
- Western Chat-Tanager, Calyptophilus tertius
- Eastern Chat-Tanager, Calyptophilus frugivorus
- Wrenthrush, Zeledonia coronata
- Green-tailed Warbler, Microligea palustris
- White-winged Warbler, Xenoligea montana
- Black-crowned Palm-Tanager / Black-crowned Tanager, Phaenicophilus palmarum
- Gray-crowned Palm-Tanager / Gray-crowned Tanager, Phaenicophilus poliocephalus
- Puerto Rican Tanager, Nesospingus speculiferus
- Jamaican Spindalis, Spindalis nigricephala
- Hispaniolan Spindalis, Spindalis dominicensis
- Puerto Rican Spindalis, Spindalis portoricensis
- Western Spindalis, Spindalis zena
Icteridae: New World Blackbirds Vigors, 1825
31 genera, 114 species HBW-16
 |
| Click for species-level tree for Icteridae |
|---|
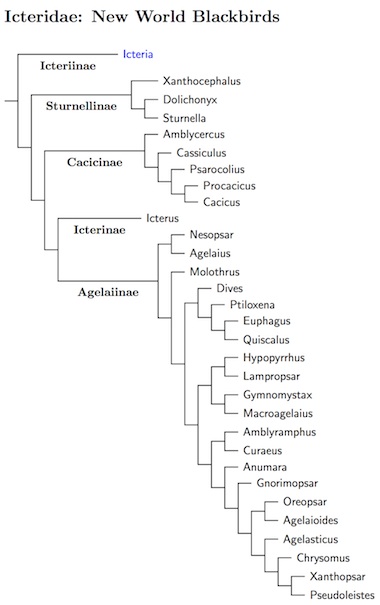
The blackbirds have not undergone quite the same taxonomic chaos that their relatives have. It's generally been clear which birds are blackbirds and which are not. However, the internal structure of the family has required modification. The major lineages were identified in Lanyon and Omland (1999), which is a fairly comprehensive genetic study of the family. Unfortunately, their results did not reveal how they fit together. The more recent studies by Price et al. (2010), Barker et al. (2013), and Powell et al. (2014) fill that gap. One issue that only Barker et al. address is whether any other species belong in the blackbird family. They examined all the genera in Icteroidae, and found that several are possibly basal blackbirds. The evidence is strongest in the case of the Yellow-breasted Chat, Icteria virens, and I have included it as a basal subfamily, Icteriinae. The two Cuban Teretistris warblers and Zeledonia are also potential basal blackbirds. Since the evidence is weaker (they could be warblers, or basal to both warblers and blackbirds), I have not assigned them to a family.
The current arrangement is primarily based on the complete analysis of Powell et al. (2014), with assists from Jacobsen and Omland (2011) and D'Horta et al. (2008) for portions of Icterus. It varies some from earlier versions primarily in the Agelaiinae. The previous interation was based on earlier work by Lanyon and Omland (1999) which included a fair amount of detail about the arrangement of species, but didn't sequence everything. For that, several more detailed papers are available. Besides the comprehensive studies of Price et al. (2010) and Powell et al. (2013), there are several focusing on specific subgroups. Omland et al. (1999) covers the Icterinae. Several papers by Price and Lanyon (2002a, b; 2004) investigate the Cacicinae. The Agelaiinae are the subject of Powell et al. (2013) and Johnson and Lanyon (1999), which Cadena et al. complete by including Hypopyrrhus. Two papers by Barker et al. clarify the species relationships within Agelaius (Barker et al. 2008a) and Sturnella (Barker et al. 2008b).
The Icteridae are divided into 5 groups. The basal group contains a single species, Yellow-breasted Chat. This had previously been considered a Parulid warbler, but seems to be an Icterid (appropriate for genus Icteria). Next comes the meadowlark group. The third group contains the caciques and oropendolas. Powell et al.'s (2014) combined analysis places the Amblycercus caciques with this group, albeit only distantly related to the others. Mitochondrial DNA puts Amblycercus sister to the blackbird/grackle group. Finally, the last two groups are the New World orioles and the blackbird/grackle group.
A paper in the Auk by Barker et al. (2008b) provides further evidence that Lilian's Meadowlark, Sturnella lilianae should be considered a separate species. When I first saw lilianae back in 1999, I was told it was a potential split. It went into my escrow list at that point. Finally, the weight of evidence favors a split, and I follow that here. AOS has since adopted the split, but used the name Chihuahan Meadowlark. Note that all the Sturnella are now called meadowlarks (SACC proposal #641)
Some reorganization of genera has been required. In the Cacicinae, the Yellow-winged Cacique, Cacicus melanicterus, is basal according to Price and Lanyon (2004), and is accordingly placed in the monotypic genus Cassiculus. They also found that Casqued Oropendola, Clypicterus oseryi, and Band-tailed Oropendola, Ocyalus latirostris, are embedded in Cacicus. Their English names have been adjusted accordingly (see SACC proposal #641). Finally, position of the Solitary Black Cacique, Cacicus solitarius, is somewhat controversial. Mitochondiral DNA says it is sister to the oropendolas, but nuclear DNA puts it next to the main cacique group. The placement here next to the other caciques follows the combined analysis of Powell et al. (2014), but we recognize the taxonomic uncertainty by placing it in its own genus Procacicus, as suggested by Fraga (2005).
The genetic data in Powell et al. (2014) provide some support for several splits in the caciques that have not been adopted (yet?) by the AOU. These include splitting:
- Yellow-billed Cacique into Prevost's Yellow-billed Cacique, Amblycercus holosericeus (including flavirostris), ranging from Mexico to Colombia (holosericeus) and the Pacific lowlands from western Colombia to northwest Peru (flavirostris), and Chapman's Yellow-billed Cacique, Amblycercus australis, of the Andean highlands from Colombia to Bolivia.
- Yellow-rumped Cacique into Western Yellow-rumped Cacique, Cacicus flavicrissus (including vitellinus), ranging from Panama, northern and central Colombia, west Ecuador and NW Peru, and Amazonian Yellow-rumped Cacique, Cacicus cela, in Amazonia.
- Scarlet-rumped Cacique into Scarlet-rumped Cacique, Cacicus microrhynchus, ranging from Honduras to most of Panama, Pacific Cacique, Cacicus pacificus, ranging from the Darien south to western Ecuador, and Subtropical Cacique, Cacicus uropygialis, in the Andes from Venezuela to southern Peru and western Bolivia.
- Mountain Cacique into Northern Mountain-Cacique, Cacicus leucoramphus (including peruvianus) ranging from Venezuela to northern Peru, and Southern Mountain-Cacique, Cacicus chrysonotus, in southern Peru and Bolivia.
The subfamily Icterinae contains only a single genus, Icterus. It's divided into three main clades, marked as subgenera of Icterus: Yphantes, Icterus, and Pendulinus.
Omland et al. (1999) found that the 4 subspecies of Icterus dominicensis fall into 3 separated groups. The group of two (dominicensis and northropi) differed by about 0.7% in DNA. The DNA separation is small enough that they could be subspecies, but large enough that they could be species. As the two are geographically separated and have distinctive plumages, I treat them as separate species. Accordingly, I split Greater Antillean Oriole into Cuban Oriole, Icterus melanopsis, Bahama Oriole, Icterus northropi, Hispaniolan Oriole, Icterus dominicensis, and Puerto Rican Oriole, Icterus portoricensis. The AOU NACC has also now endorsed the split. Interestingly, the Greater Antillean Oriole was a fairly recent split from Black-cowled Oriole. The arrangement of these species follows Sturge et al. (2009), which has better taxon sampling.
Jacobsen et al.'s (2010) analysis of the Z-chromosome yields similar results, except to introduce some uncertainty concerning the exact placement of the Bar-winged Oriole, Icterus maculialatus, and the Baltimore, Bullock's, and Black-backed Orioles (galbula, bullockii, and abeillei). The current arrangement is based on a follow-up paper by Jacobsen et al. (2011).
The Ochre Oriole, Icterus fuertesi, is also weakly, but distinctly, genetically separated (0.6%) from Orchard Oriole, Icterus spurius. These are also geographically isolated and have distinctive plumage. To maintain a consistent standard, I also consider these separate species. There are other potential oriole splits that I don't have sufficient information on.
Omland et al. (1999) also found that the Moriche Oriole, Icterus chrysocephalus is nested in the Epaulet Orioles. This whole complex has recently been studied by D'Horta et al. (2008). Although it has previously been divided into 6 subspecies, they found 4 phylogenetic species (subspecies) in two hybridizing clades, cayanensis/chrysocephalus and pyrrhopterus/tibialis. The subspecies valenciobuenoi appears to be a pyrrhopterus-tibialis hybrid while periporphyrus belongs to pyrrhopterus. There doesn't seem to be any evidence of recent interbreeding between the two groups, suggesting they are separate biological species. I recognize them here as Epaulet Oriole, Icterus cayanensis (including chrysocephalus) and Variable Oriole, Icterus pyrrhopterus (including tibialis). Although the name “Yellow-shouldered Oriole” has previously been used for pyrrhopterus (e.g., Sibley and Monroe, 1993), it doesn't describe the bird well as the epaulet color varies (pyrrhopterus itself has chestnut epaulets, the subspecies tibialis does have yellow epaulets). The SACC adopted the name Variable Oriole (see SACC proposal #418).
That brings us to the blackbird/cowbird/grackle group, Agelaiinae. The grackles were studied by Lanyon and Omland (1999), DaCosta et al. (2008), and Powell et al. (2008), and all of the current species have been sequenced. There are four groups: (1) a basal group containing the Agelaius blackbirds, (2) the Molothrus cowbirds, (3) a blackbird/Quiscalus grackle group, and (4) the remaining blackbirds and grackles.
Powell et al. (2014) found that the Cuban Blackbird is not sister to the two Dives blackbirds, and has been moved to the montoypic genus Ptiloxena (Chapman 1892) to reflect this.
Both DaCosta et al. and Powell et al. found that the Great-tailed Grackle complex contains two groups, and that the Boat-tailed Grackle is sister to the eastern Great-tailed Grackles. Powell et al. also found that the Slender-billed Grackle was sister to the western Great-tailed Grackles. The two Great-tailed Grackle groups seem to interbreed freely where they meet in the southwestern US, and so constitute a biological species, even though they are a paraphyletic group. What may have happened is that the Great-tailed Grackles split into two groups. Each furthered separated into two more groups (E and W Great-tailed Grackles and palustris and major). Eventually, the E and W Great-tailed Grackles came back into contact and started interbreeding, while palustris and major set up barriers to interbreeding with the Great-tailed Grackles. I've left these three grackle species in a trichotomy because of their complex relationship. Powell et al. (2008) also note that the Carib Grackle seems to include more than one species. How many more is not yet known, but based on Powell et al. (2014), I've split Barbados Grackle, Quiscalus fortirostris (including contrusus) from Carib Grackle, Quiscalus lugubris.
Forbes's Blackbird is not sister to the Austral Blackbird and has been placed in the monotypic genus Anumara (Powell et al., 2014).
The Bay-winged Cowbird, Agelaioides badius, is split into Grayish Baywing, Agelaioides badius, and Pale Baywing, Agelaioides fringillarius. (SACC Proposal #642)
Icteriinae: Yellow-breasted Chat Baird, 1858
- Yellow-breasted Chat, Icteria virens
Sturnellinae: Meadowlarks Chenu & des Murs, 1853
- Yellow-headed Blackbird, Xanthocephalus xanthocephalus
- Bobolink, Dolichonyx oryzivorus
- Western Meadowlark, Sturnella neglecta
- Eastern Meadowlark, Sturnella magna
- Lilian's Meadowlark / Chihuahan Meadowlark, Sturnella lilianae
- Red-breasted Meadowlark / Red-breasted Blackbird, Sturnella militaris
- White-browed Meadowlark / White-browed Blackbird, Sturnella superciliaris
- Peruvian Meadowlark, Sturnella bellicosa
- Long-tailed Meadowlark, Sturnella loyca
- Pampas Meadowlark, Sturnella defilippii
Cacicinae: Caciques and Oropendolas Bonaparte, 1853, emended Ridgway, 1902
- Prevost's Yellow-billed Cacique, Amblycercus holosericeus
- Chapman's Yellow-billed Cacique, Amblycercus australis
- Yellow-winged Cacique / Mexican Cacique, Cassiculus melanicterus
- Chestnut-headed Oropendola, Psarocolius wagleri
- Russet-backed Oropendola, Psarocolius angustifrons
- Dusky-green Oropendola, Psarocolius atrovirens
- Crested Oropendola, Psarocolius decumanus
- Green Oropendola, Psarocolius viridis
- Olive Oropendola, Psarocolius bifasciatus
- Montezuma Oropendola, Psarocolius montezuma
- Black Oropendola, Psarocolius guatimozinus
- Baudo Oropendola, Psarocolius cassini
- Solitary Black Cacique / Solitary Cacique, Procacicus solitarius
- Golden-winged Cacique, Cacicus chrysopterus
- Ecuadorian Cacique, Cacicus sclateri
- Selva Cacique, Cacicus koepckeae
- Western Yellow-rumped Cacique, Cacicus flavicrissus
- Amazonian Yellow-rumped Cacique, Cacicus cela
- Scarlet-rumped Cacique, Cacicus microrhynchus
- Pacific Cacique, Cacicus pacificus
- Subtropical Cacique, Cacicus uropygialis
- Northern Mountain-Cacique, Cacicus leucoramphus
- Southern Mountain-Cacique, Cacicus chrysonotus
- Band-tailed Cacique / Band-tailed Oropendola, Cacicus latirostris
- Red-rumped Cacique, Cacicus haemorrhous
- Casqued Cacique / Casqued Oropendola, Cacicus oseryi
Icterinae: Orioles Vigors, 1825
- Scott's Oriole, Icterus parisorum
- Audubon's Oriole, Icterus graduacauda
- Yellow-backed Oriole, Icterus chrysater
- Baltimore Oriole, Icterus galbula
- Bullock's Oriole, Icterus bullockii
- Black-backed Oriole, Icterus abeillei
- Streak-backed Oriole, Icterus pustulatus
- Jamaican Oriole, Icterus leucopteryx
- Orange Oriole, Icterus auratus
- Yellow Oriole, Icterus nigrogularis
- Altamira Oriole, Icterus gularis
- Yellow-tailed Oriole, Icterus mesomelas
- Spot-breasted Oriole, Icterus pectoralis
- White-edged Oriole, Icterus graceannae
- Campo Troupial, Icterus jamacaii
- Venezuelan Troupial, Icterus icterus
- Orange-backed Troupial, Icterus croconotus
- Bar-winged Oriole, Icterus maculialatus
- Black-vented Oriole, Icterus wagleri
- Hooded Oriole, Icterus cucullatus
- Black-cowled Oriole, Icterus prosthemelas
- Orchard Oriole, Icterus spurius
- Ochre Oriole, Icterus fuertesi
- Cuban Oriole, Icterus melanopsis
- Bahama Oriole, Icterus northropi
- Martinique Oriole, Icterus bonana
- Puerto Rican Oriole, Icterus portoricensis
- Montserrat Oriole, Icterus oberi
- Hispaniolan Oriole, Icterus dominicensis
- St. Lucia Oriole, Icterus laudabilis
- Orange-crowned Oriole, Icterus auricapillus
- Epaulet Oriole, Icterus cayanensis
- Variable Oriole, Icterus pyrrhopterus
Agelaiinae: Blackbirds, Cowbirds, and Grackles Swainson, 1831
- Jamaican Blackbird, Nesopsar nigerrimus
- Tawny-shouldered Blackbird, Agelaius humeralis
- Yellow-shouldered Blackbird, Agelaius xanthomus
- Tricolored Blackbird, Agelaius tricolor
- Red-winged Blackbird, Agelaius phoeniceus
- Red-shouldered Blackbird, Agelaius assimilis
- Screaming Cowbird, Molothrus rufoaxillaris
- Giant Cowbird, Molothrus oryzivorus
- Shiny Cowbird, Molothrus bonariensis
- Brown-headed Cowbird, Molothrus ater
- Bronzed Cowbird, Molothrus aeneus
- Melodious Blackbird, Dives dives
- Scrub Blackbird, Dives warczewiczi
- Cuban Blackbird, Ptiloxena atroviolacea
- Rusty Blackbird, Euphagus carolinus
- Brewer's Blackbird, Euphagus cyanocephalus
- Common Grackle, Quiscalus quiscula
- Nicaraguan Grackle, Quiscalus nicaraguensis
- Carib Grackle, Quiscalus lugubris
- Barbados Grackle, Quiscalus fortirostris
- Greater Antillean Grackle, Quiscalus niger
- Boat-tailed Grackle, Quiscalus major
- Great-tailed Grackle, Quiscalus mexicanus
- Slender-billed Grackle, Quiscalus palustris
- Red-bellied Grackle, Hypopyrrhus pyrohypogaster
- Velvet-fronted Grackle, Lampropsar tanagrinus
- Oriole Blackbird, Gymnomystax mexicanus
- Mountain Grackle / Colombian Mountain-Grackle, Macroagelaius subalaris
- Golden-tufted Grackle / Golden-tufted Mountain-Grackle, Macroagelaius imthurni
- Scarlet-headed Blackbird, Amblyramphus holosericeus
- Austral Blackbird, Curaeus curaeus
- Forbes's Blackbird, Anumara forbesi
- Chopi Blackbird, Gnorimopsar chopi
- Bolivian Blackbird, Oreopsar bolivianus
- Grayish Baywing, Agelaioides badius
- Pale Baywing, Agelaioides fringillarius
- Yellow-winged Blackbird, Agelasticus thilius
- Pale-eyed Blackbird, Agelasticus xanthophthalmus
- Unicolored Blackbird, Agelasticus cyanopus
- Yellow-hooded Blackbird, Chrysomus icterocephalus
- Chestnut-capped Blackbird, Chrysomus ruficapillus
- Saffron-cowled Blackbird, Xanthopsar flavus
- Yellow-rumped Marshbird, Pseudoleistes guirahuro
- Brown-and-yellow Marshbird, Pseudoleistes virescens
Parulidae: Wood-Warblers Wetmore et al., 1947 (1831)
21 genera, 118 species HBW-15
Before getting to the warblers themselves, there are some non-warbler genera to consider. One is Peucedramus, the Olive Warbler, which is now placed in its own monotypic family near the accentors (Prunellidae). The other genus is Granatellus. Klicka et al. (2007) show that the Granatellus chats belong to the Cardinalidae. Other taxa that have been considered warblers, and may actually be warblers are the Yellow-breasted Chat (Icteria), the two Teretistris “warblers”, and the Wrenthrush (Zeledonia). Current evidence from Barker et al. (2013) indicates these near-warblers are outside Parulidae proper. However, the support for this is not great, and it is possible, albeit unlikely, that will turn out to be Parulidae after all.
Now that Parulidae has been reduced to the core wood-warblers, we have a pretty good understanding of its phylogeny.
 |
| Click for Parulidae tree |
|---|
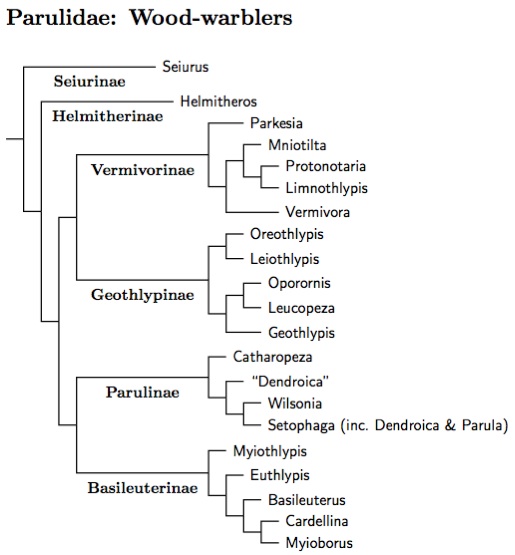
The division of Parulidae into subfamilies is based on Lovette et al. (2010). See also Lovette and Bermingham (2002), Klein et al. (2004), and Lovette and Hochachka (2006). I've relied more heavily on Lovette et al. (2010) as it analyzes more data from more genes. Be warned that the position of some taxa is not stable in these papers, which means it's not reliable here either. In particular, the subfamilies may fit together slightly differently.
There are two basal species. First, the Ovenbird, which is not closely related to the waterthrushes after all (they have been moved to Vermivorinae, in the new genus Parkesia defined by Sangster, 2008b). Next in sequence is the Worm-eating Warbler. The remaining warblers are then divided into 4 groups, here called Vermivorinae, Geothlypinae, Parulinae, and Basileuterinae. The balance of the evidence suggests that Vermivorinae is sister to Geothlypinae and that Parulinae and Basileuterinae are sisters.
Seiurinae Baird, 1864 (1862)
- Ovenbird, Seiurus aurocapilla
Helmitherinae Bonaparte, 1853
- Worm-eating Warbler, Helmitheros vermivorum
Vermivorinae Swainson, 1831
Lovette et al. (2010) consider Parkesia basal in Vermivorinae, although support could be stronger. There seems to be consensus among the various analyses that Protonotaria and Limnothlypis are sisters. Mniotilta is another taxon that moves around, but Lovette et al. (2010) placed it sister to the other two. There are similar issues involving Vermivora. Note also that Vermivora has been divided into two pieces: the three true Vermivora and Leiothlypis.
Like the Ovenbird and Worm-eating Warbler, the Vermivorinae all breed in North America, and except for the Black-and-white Warbler, and their winter grounds center on Central America and the West Indies, including the Caribbean coast of South America. Although the species may have changed a bit, these genera likely predate the joining of the Americas.
- Northern Waterthrush, Parkesia noveboracensis
- Louisiana Waterthrush, Parkesia motacilla
- Black-and-white Warbler, Mniotilta varia
- Prothonotary Warbler, Protonotaria citrea
- Swainson's Warbler, Limnothlypis swainsonii
- Bachman's Warbler, Vermivora bachmanii
- Golden-winged Warbler, Vermivora chrysoptera
- Blue-winged Warbler, Vermivora cyanoptera
Geothlypinae Baird, 1858
This version of Geothlypinae is based on Lovette et al. (2010). It is similar to the results in Lovette and Hochachka (2006). Klein et al. (2004) put the two parts of Geothlypinae in different places, but with low support. One piece of Geothlypinae is restricted to Middle and North America. It consists of Oreothlypis (formerly part of of Parula) and the dull Vermivora, now called Leiothlypis (Sangster, 2008a).
The AOU NACC has voted to treat Leiothlypis and Oreothlypis as a single genus (Oreothlypis). However, I agree wtth the committee members who stressed the difference between the dull, mostly Nearctic, Leiothlypis and the colorful Neotropical Oreothlypis, and so keep them in separate genera. More recently, the SACC voted to maintain the distinction between Leiothlypis and Oreothlypis.
Although the position of this clade is somewhat uncertain, the arrangement of species inside it is pretty good. The main issue concerns the Nashville Warbler, which shows up in two different places in Zink et al. (2000). The western race ridgwayi (Calaveras Warbler) appears to be sister to Lucy's Warbler and the eastern race ruficapilla appears to be basal to a clade that additionally contains Virginia's Warbler. Weir and Schluter (2004) is consistent with this. However, Lovette et al. (2010), using more genes, found the that ridgwayi (the only race of Nashville Warbler that they include) is sister to Virginia's, not Lucy's. Right now, the sample size is too small and the various results too inconsistent to draw any conclusion. Further research will be needed to clarify the situation.
The other portion of Geothlypinae consists of Oporornis and Geothlypis. Although Oporornis breeds in North American, the Geothlypis yellowthroats have moved into South America as well as North.
Geothlypis and Oporornis have sometimes been thought congeneric. Lovette and Bermingham (2002) and Lovette and Hochachka (2006) found Geothlypis nested within Oporornis. Escalante et al. (2009) helps clarify the situation, and suggests calling all of them Geothlypis as the best solution.
Due to the shape of the tree, the only one of these that can take the name Oporornis is the type species, the Connecticut Warbler. The Kentucky Warbler looks more like a yellowthroat than it does like Connecticut, so I'm happy enough including it in Geothlypis. Some plumages of MacGillivray's and Mourning are tough to distinguish from yellowthroats, so I'm not opposed to rolling them in too. The Connecticut is more distinctive. Besides, it walks! So for now, I'm leaving it in Oporornis.
Although Semper's Warbler (Leucopeza) of St. Lucia is usually considered close to Teretistris, Lovette et al. (2010) found that it belongs in the Oporornis-Geothlypis clade. I've used their preferred tree which has Leucopeza sister to Oporornis, but while Leucopeza is certaintly relatively basal, other arrangements have not been entirely ruled out. In any event, it continues the West Indian theme.
There are some other changes based on Escalante et al. (2009) too. The Chiriqui Yellowthroat, Geothlypis chiriquensis, Black-lored Yellowthroat, Geothlypis auricularis, and Southern Yellowthroat, Geothlypis velata (including peruviana), are all split from Masked Yellowthroat, Geothlypis aequinoctialis. The SACC had previously rejected this split, the case is now stronger. They had pointed to greater geographic variation in the Common Yellowthroat, but Common Yellowthroat has a more or less continuous range, while these taxa are separated. If they were on islands, there would be no question about splitting them, and I think the same rule should apply. The case of the Chiriqui Yellowthroat is different. It is not really part of the aequinoctialis group, being more closely related to semiflava. We could merge it into semiflava, but it seems too distinct for that and has been considered a separate species. The Central American race of semiflava, bairdi is actually closer to chiriquensis than to South American semiflava, so it gets split too. The name Baird's Yellowthroat has previously been applied to Geothlypis bairdi and I use it here.
- Crescent-chested Warbler, Oreothlypis superciliosa
- Flame-throated Warbler, Oreothlypis gutturalis
- Tennessee Warbler, Leiothlypis peregrina
- Orange-crowned Warbler, Leiothlypis celata
- Colima Warbler, Leiothlypis crissalis
- Lucy's Warbler, Leiothlypis luciae
- Nashville Warbler, Leiothlypis ruficapilla
- Virginia's Warbler, Leiothlypis virginiae
- Connecticut Warbler, Oporornis agilis
- Semper's Warbler, Leucopeza semperi
- MacGillivray's Warbler, Geothlypis tolmiei
- Mourning Warbler, Geothlypis philadelphia
- Kentucky Warbler, Geothlypis formosa
- Gray-crowned Yellowthroat, Geothlypis poliocephala
- Masked Yellowthroat, Geothlypis aequinoctialis
- Black-lored Yellowthroat, Geothlypis auricularis
- Southern Yellowthroat, Geothlypis velata
- Black-polled Yellowthroat, Geothlypis speciosa
- Olive-crowned Yellowthroat, Geothlypis semiflava
- Baird's Yellowthroat, Geothlypis bairdi
- Chiriqui Yellowthroat, Geothlypis chiriquensis
- Belding's Yellowthroat, Geothlypis beldingi
- Bahama Yellowthroat, Geothlypis rostrata
- Common Yellowthroat, Geothlypis trichas
- Altamira Yellowthroat, Geothlypis flavovelata
- Hooded Yellowthroat, Geothlypis nelsoni
Parulinae Wetmore et al., 1947 (1831)
The rest of the wood-warblers break into two groups: Parulinae and Basileuterinae. The Parulinae is the smaller group, and so is placed ahead of Basileuterinae. Most Parulinae are usually placed in the genus Dendroica, which has recently been studied by Rabosky and Lovette (2008). The treatment here is based on Lovette et al. (2010), which is generally congruent with their results. Lovette and Bermingham (1999), Lovette et al. (1999), Klein et al. (2004), and Lovette and Hochachka (2006) were also consulted.
Although most of the Parulinae are North American breeders with winter ranges of some extending well into South America, the basal species are West Indian. These three "Dendroica" may group together, as shown here, but support is not strong. However, there is other support—they also all have a distinctive first-year plumage and reputedly have similar tail flicking behavior. These really should be in a different genus from the other Parulinae. Unless almost all the Parulinae are put in one genus, as the AOU's NACC has now done, this group will need a new genus name.
The core Dendroica start at point A. There are five clades. The clade at B includes the two Parulas. If we don't want to merge the Dendroica (G.R. Gray 1842) into Parula (Bonaparte 1838), or both into Setophaga (Swainson 1827), kirtlandi, tigrina and cerulea can be moved to Parula. Alternative treatments would be to merge Dendroica into Parula, or to name the clades as genera or subgenera as this would better represent the phylogenetic tree. In the latter case, names would be needed for clades C, E, and the Black-throated Blue Warbler (in a clade by itself).
The AOU has chosen to put almost all of Parulinae in Setophaga. I think that is a mistake, but one that is forced by the lack of an available name for the three "Dendroica", which seem rather different from the birds in clade A. Indeed, the Hooded Warbler also seems different, and I have left it in Wilsonia (contra AOU). I would actually prefer to keep the American Redstart separate also, but feel that would make the checklist too different from the AOU checklist.
The 43 recognized subspecies of Yellow Warbler, Setophaga petechia, (Browning, 1994) have sometimes been divided into as many as three species (Yellow, Golden, Mangrove). The paper by Klein and Brown (1994) threw cold water on the idea of three species as their genetic results found that while most of the Mangrove and Golden races were mixed together in a clade, it did not split into Mangrove and Golden groups. They were well-mixed. Even the two species notion did not fare well because a sample of one of the west coast Mangrove races (apparently an adult male castaneiceps) appeared to be more closely related to the northern group than to the other Golden/Mangrove races. Support for this was weak, and while it did not rule out the two-species treatment, it certainly did not support it.
Milot et al. (2000) focused on the most northern races (Canada and Alaska). Analysis of part of the control region of 155 birds revealed some geographic structure. They found 60 haplotypes. The western haplotypes formed a clade, as did most of the eastern haplotypes. However, some eastern haplotypes were closer to the western group. Boulet and Gibbs (2006) expanded the Milot et al. dataset to 684 Yellow Warblers across the US and Canada. They also found that the northern Yellow Warblers divided into two clades: an eastern clades and a mixed clade containing a western subclade. Based on range, the eastern birds correspond to aestiva and amnicola while the western birds are banksi, rubiginosa, brewsteri, morcomi, and probably parkesi (its range was not well-sampled).
Boulet and Gibbs (2006) found that the northern Yellow Warblers had a sister southern clade. These included the few Golden/Mangrove Warblers sampled, together with 30-35 individuals from the southwest, most from Lake Mead, NV and Flagstaff, AZ, with a few as far north as Vernal, UT. This indicates that sonorana (and probably the unsampled dugesi) belong in the southern clade with the Golden/Mangrove warblers. Comparison with Klein and Brown (2004) suggests that all of their Golden/Mangrove warblers (except possibly the single castaneiceps) belong in the southern clade. The fact that at least one, probably two, of the northern Yellow Warblers belong in the southern clade is evidence against treating the Golden/Mangrove Warblers as a separate species.
That is not the end of the story. Browne et al. (2008) examined Yellow Warblers from the Galapagos and Cocos Islands (subspecies aureola). They found a lack of gene flow between them and the mainland samples, which raises the question of whether they are a separate biological species. They compared the aureola haplotypes with 55 previously published haplotypes, purportedly from Milot et al. (2000). However, this included 5 haplotypes from Pennsylvannia that cannot have come from Milot et al. (2000). They may have come from Boulet and Gibbs (2006) and Boulet et al. (2006), but it is not clear. Oddly, with the possible exception of a single bird from somewhere in Costa Rica, they did not include any Pacific coast Mangrove Warblers in their analysis, not even the races jubaris and peruviana which are both geographically closest (Darié to northern Peru) and similar in appearance to aureola (rufous-capped rather than rufous-hooded).
They also carried out some measurements of genetic distance. Not surprisingly, auerola was much closer to the Golden/Mangrove Warblers than to the northern Yellow Warblers. Exactly how close is hard to say (see Boulet and Gibbs, 2006), but this also gives support for retaining the Golden/Mangrove Warblers as subspecies of Yellow Warbler.
The Yellow Warbler is not the only wood-warbler that may include multiple species. There are also issues with the Yellow-rumped Warbler, Setophaga coronata, but perhaps not just the ones you think. Milá et al. (2007) examined seven Yellow-rumped Warbler populations. They found that two of the southern races seem to be pretty isolated genetically speaking. In short, there is a good case for considering the allopatric Mexican and Guatemalan populations to be separate species: Black-fronted Warbler, Setophaga nigrifrons, and Goldman's Warbler, Setophaga goldmani. Milá et al.'s work left a question mark concerning the population from the Chiricahua and Huachuca mountains (memorabilis), as it is traditionally considered part of nigrifrons.
As for the Myrtle/Audubon's split, Milá et al. found evidence of fairly recent gene flow, arguing against species status. However, Brelsford and Irwin (2009) carefully examined a portion of the hybrid zone in Canada, and showed there are now substantial barriers to gene flow. This is strong evidence that the two are separate biological species, even though the separation between them is recent and genetically incomplete. As a result, I have also split them into the familiar Myrtle Warbler, Setophaga coronata, and Audubon's Warbler, Setophaga auduboni.
This is not the end of the story! Brelsford et al. (2011) argues that Audubon's Warbler actually formed as a hybrid between the Myrtle and Black-fronted Warblers. This is particularly interesting as there are few documented cases of this sort of speciation among birds. The complications continue in Milá et al. (2011), where they find surprising genetic differences between northern and southern populations of Audubon's Warbler, with the northern closer to Myrtle and southern closer to Black-fronted. Whether the four (or five!) types of yellow-rumped warblers are best treated as 1, 2, 3, or 4 species is currently unclear. After reconsidering the evidence, the differences between the northern and southern Audubon's and Black-fronted Warblers seem more clinal, and I currently treat them as a single species, Audubon's Warbler, but treat Goldman's Warbler as a separate species. This also seems consistent with the recent paper by Toews et al. (2014).
Based on McKay et al. (2010a), I've split the Bahama Warbler, Setophaga flavescens, from the Yellow-throated Warbler, Setophaga dominica. These have long been candidates for a split as the Bahama Warbler is quite distinctive. McKay et al. provide new evidence that there are barriers to hybridization with the closely related Yellow-throated Warbler, even though the genetic differences are quite small indicating a fairly recent divergence.
- Whistling Warbler, Catharopeza bishopi
- Plumbeous Warbler, "Dendroica" plumbea
- Elfin-woods Warbler, "Dendroica" angelae
- Arrowhead Warbler, "Dendroica" pharetra
- Hooded Warbler, Wilsonia citrina
- American Redstart, Setophaga ruticilla
- Kirtland's Warbler, Setophaga kirtlandii
- Cape May Warbler, Setophaga tigrina
- Cerulean Warbler, Setophaga cerulea
- Northern Parula, Setophaga americana
- Tropical Parula, Setophaga pitiayumi
- Magnolia Warbler, Setophaga magnolia
- Bay-breasted Warbler, Setophaga castanea
- Blackburnian Warbler, Setophaga fusca
- Yellow Warbler / American Yellow Warbler, Setophaga petechia
- Blackpoll Warbler, Setophaga striata
- Chestnut-sided Warbler, Setophaga pensylvanica
- Black-throated Blue Warbler, Setophaga caerulescens
- Palm Warbler, Setophaga palmarum
- Pine Warbler, Setophaga pinus
- Olive-capped Warbler, Setophaga pityophila
- Yellow-throated Warbler, Setophaga dominica
- Bahama Warbler, Setophaga flavescens
- Myrtle Warbler, Setophaga coronata
- Audubon's Warbler, Setophaga auduboni
- Goldman's Warbler, Setophaga goldmani
- Prairie Warbler, Setophaga discolor
- Vitelline Warbler, Setophaga vitellina
- Adelaide's Warbler, Setophaga adelaidae
- Barbuda Warbler, Setophaga subita
- St. Lucia Warbler, Setophaga delicata
- Black-throated Gray Warbler, Setophaga nigrescens
- Grace's Warbler, Setophaga graciae
- Townsend's Warbler, Setophaga townsendi
- Hermit Warbler, Setophaga occidentalis
- Black-throated Green Warbler, Setophaga virens
- Golden-cheeked Warbler, Setophaga chrysoparia
Basileuterinae Informal
The Basileuterinae are the warblers that moved into South America in a big way. Myiothlypis (which includes Phaeothlypis) is the basal group. Note that Myiothlypis (Cabanis 1850, type nigrocristata) has priority over Phaeothlypis (Todd 1929, type fulvicauda). Gutiérrez-Pinto et al. (2012) found that the Santa Marta Warbler, formerly Basileuterus basilicus, belongs in Myiothlypis as Myiothlypis basilica.
The other clade has one branch consisting of Euthlypis and the remaining Basileuterus, and a more heterogeneous branch. Both the basal clade and the Basileuterus branch are strongly South American in distribution. Some of the species here need further study. Lovette (2004) examined the Riverbank/Buff-rumped Warbler complex without coming to any firm conclusion about which races belong to which warbler, or whether they should all be lumped into one or split into as many as six species.
The Roraiman Warbler, Basileuterus roraimae, is split from Two-banded Warbler, Basileuterus bivittatus. Hilty (2003) had recommended this split, and Lovette et al. (2010) show that there is substantial genetic distance between the two taxa.
The White-bellied Warbler, Basileuterus hypoleucus, is no longer considered a separate species, but rather a color morph of Golden-crowned Warbler, Basileuterus culicivorus. It had previously been suspected that they were conspecific, and Vilaça and dos Santos (2010) have found substantial genetic evidence of it.
The Three-striped Warbler, Basileuterus tristriatus, has been split into four species based on Donegan (2014a) and Gutiérrez-Pinto et al. (2012).
- Black-eared Warbler, Basileuterus melanotis, including chitrensis.
- Tacarcuna Warbler, Basileuterus tacarcunae (monotypic).
- Three-striped Warbler, Basileuterus tristriatus, including pariae, bessereri, meridanus, auricularis, daedalus, baezae, and sanlucasensis, as well as an undescribed form from southern Peru.
- Yungas Warbler, Basileuterus punctipectus, including inconspicuus and canens.
Both Pérez-Emán (2005) and Lovette et al. (2010) have examined the rest of the Basileuterinae. They consist of two parts, a North and Middle American group including two former Wilsonia (Wilson's and Canada Warblers), and the Myioborus whitestarts. (Although I follow AOU in making redstart the primary name in this list, I am not happy with it and much prefer whitestart as used by Curson et al. (1994), Ridgely and Greenfield (2001), and Hilty (2003).)
At first this seemed a strange pairing, but it's really not too hard to imagine kinship between the Canada Warbler and Spectacled Whitestart, or for that matter between Wilson's and Red-faced Warbler, once you look past the color scheme. I've taken Lovette's et al.'s advice to merge Ergaticus into Cardellina, along with the two ex-Wilsonia.
Within Myioborus, the Painted Redstart is basal, and the lower elevation Slate-throated Redstart is basal among the rest. This suggests a separate movement into South America for Myioborus. The remaining Myioborus are mainly higher-elevation species. The higher elevation areas are separated, and the species differentiation reflects this. There are some issues concerning species boundaries: whether or not to lump ornatus and melanocephalus, and whether or not to lump or partially lump the Tepui clade. If ornatus and melanocephalus are kept separate, the Ecuadorian subspecies M. m. ruficoronatus should be moved to ornatus.
- Roraiman Warbler, Myiothlypis roraimae
- Two-banded Warbler, Myiothlypis bivittata
- Golden-bellied Warbler / Cuzco Warbler, Myiothlypis chrysogaster
- White-lored Warbler, Myiothlypis conspicillata
- Gray-throated Warbler, Myiothlypis cinereicollis
- Russet-crowned Warbler, Myiothlypis coronata
- Gray-and-gold Warbler, Myiothlypis fraseri
- Santa Marta Warbler, Myiothlypis basilica
- Citrine Warbler, Myiothlypis luteoviridis
- White-striped Warbler, Myiothlypis leucophrys
- Flavescent Warbler, Myiothlypis flaveola
- White-browed Warbler / White-rimmed Warbler, Myiothlypis leucoblephara
- Gray-headed Warbler, Myiothlypis griseiceps
- Buff-rumped Warbler, Myiothlypis fulvicauda
- Riverbank Warbler, Myiothlypis rivularis
- Black-crested Warbler, Myiothlypis nigrocristata
- Pale-legged Warbler, Myiothlypis signata
- Fan-tailed Warbler, Euthlypis lachrymosa
- Rufous-capped Warbler, Basileuterus rufifrons
- Golden-browed Warbler, Basileuterus belli
- Black-cheeked Warbler, Basileuterus melanogenys
- Pirre Warbler, Basileuterus ignotus
- Golden-crowned Warbler, Basileuterus culicivorus
- Black-eared Warbler, Basileuterus melanotis
- Tacarcuna Warbler, Basileuterus tacarcunae
- Three-banded Warbler, Basileuterus trifasciatus
- Three-striped Warbler, Basileuterus tristriatus
- Yungas Warbler, Basileuterus punctipectus
- Canada Warbler, Cardellina canadensis
- Wilson's Warbler, Cardellina pusilla
- Red-faced Warbler, Cardellina rubrifrons
- Red Warbler, Cardellina rubra
- Pink-headed Warbler, Cardellina versicolor
- Painted Redstart / Painted Whitestart, Myioborus pictus
- Slate-throated Redstart / Slate-throated Whitestart, Myioborus miniatus
- Brown-capped Redstart / Brown-capped Whitestart, Myioborus brunniceps
- Yellow-crowned Redstart / Yellow-crowned Whitestart, Myioborus flavivertex
- White-fronted Redstart / White-fronted Whitestart, Myioborus albifrons
- Golden-fronted Redstart / Golden-fronted Whitestart, Myioborus ornatus
- Spectacled Redstart / Spectacled Whitestart, Myioborus melanocephalus
- Collared Redstart / Collared Whitestart, Myioborus torquatus
- Paria Redstart / Paria Whitestart, Myioborus pariae
- White-faced Redstart / White-faced Whitestart, Myioborus albifacies
- Saffron-breasted Redstart / Guaiquinima Whitestart, Myioborus cardonai
- Tepui Redstart / Tepui Whitestart, Myioborus castaneocapilla