Corvoidea
 |
| Click for Corvoidea genus tree |
|---|
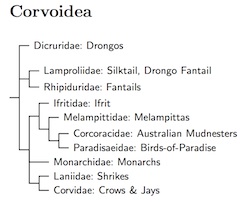
The Corvoidea form the inner core of the corvids. They are more closely related to each other than to anything else. Except perhaps for an isolated species or two, there's not any real question about which families, or even genera, belong to this core group. However, the ordering of the families has been rather obscure. This has improved with the comprehensive six-gene analysis of Jønsson et al. (2011b), especially figure S1-B. This mostly resolves the Corvoidea, but support for some of the groupings is still a bit shaky. Moyle et al. (2016) did not lead to any further changes. The key possible contribution concerning the position of the birds-of-paradise (Paradisaeidae), but their analysis was not only equivocal, but lacked some of the key taxa.
Jønsson et al. (2008b) put the Crested Jay near the shrikes. In contrast, Jønsson et al. (2011b) show two alternative possibilities. They show on its own basal branch in figure S1-B, but nest it further inside the Corvoidea in S1-A. However, Aggerbeck et al. (2014) put it in the same clade as Lanius, with strong support.
The fantails (Rhipiduridae) are the basal group in the Corvoidea, followed by the drongos (Dicruridae) are the basal group. There's some chance that the other order is correct. The remaining species split into two clades: with the Ifrit (Ifritidae), melampittas (Melampittidae), mudnesters (Corcoracidae), and birds-of-paradise (Paradisaeidae) on one side and the monarchs (Monarchidae), shrikes (Laniidae), and jays and crows (Corvidae) on the other.
There is another interesting small change in the Corvidae besides the removal of the Crested Jay. The other ringer is Hume's Groundpecker (or Ground Jay), Pseudopodoces humilis. It was thought to be a corvid, the smallest of them. It is so listed even in the Howard and Moore list (Dickinson 2003), but genetic evidence (James et al., 2003; Gill et al., 2005; Tietze et al., 2012) shows it is actually a parid!
Lamproliidae: Silktail, Drongo Fantail Wolters, 1977
3 genera, 3 species Not HBW Family (HBW-11:322, 14:206)
Irestedt et al. (2008) found that the Silktail is not a monarch (or anything else it had been considered in the past). Rather, it is most closely related to the fantails, particularly the Drongo Fantail (formerly Pygmy Drongo, but not a drongo and not really a proper fantail). These species closest relatives are the fantails. Even the fantails are only distant relatives. Jønsson et al. (2016) estimate the common ancestor at about 22 million years ago, making it reasonable to put them in their own family. In constrast, the most recent common ancestor of the fantails proper seems to have been about 15 million years ago.
Surprisingly, Jønsson et al. (2018) found that the previously unsequenced Cerulean Paradise-Flycatcher, Eutrichomyias rowleyi is more closely related to the silktails than to anything else. Although J&soslash; et al. put the Lamproliidae/Rhipiduridae split at about 18mya, Moyle et al. (2016), which I am generally following, place it at about 13 mya, consistent with including it as the basal member of the silktail family rather than separating it as a monotypic family.
- Cerulean Paradise-Flycatcher, Eutrichomyias rowleyi
- Drongo Fantail, Chaetorhynchus papuensis
- Silktail, Lamprolia victoriae
Rhipiduridae: Fantails Sundevall, 1872
8 genera, 48 species HBW-11
 |
| Click for Fantail species tree |
|---|
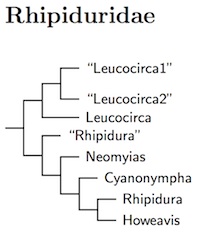
The Yellow-bellied Fantail, formerly Rhipidura hypoxantha, has been moved as it is not a fantail (Nyári et al., 2009). It now takes the scientific name Chelidorhynx hypoxanthus and is one of the Stenostiridae. The overall arrangement of the fantails is based on the Bayesian analysis in Nyári et al. (2009).
Based on Nyári et al. (2009) and Jønsson et al. (2016), I have split Rhipidura into 8 genera. Three do not seem to have available names, so I have given them temporary designations. The first group is a single bird from the Lesser Sundas, Brown-capped Fantail, which I'm calling genus "Leucocirca1". This is followed by the Thicket-Fantails of New Guinea, which also don't have a name. They become "Leucocirca2". The true Leucocirca (Swainson 1838, type leucophrys) range from India to Australia. The Black Fantail of New Guinea gets its own temporary genus "Rhipidura". Next comes Neomyias (Sharpe 1879, type euryurus). This genus ranges from Malaysia to Australia. After that is the Philippine genus Cyanonympha (Oberholser 1911, type superciliaris). Finally, we get to the true Rhipidura (Vigors and Horsfield 1827, type fulignosa), the gray and streaked fantails. They range from Australo-Papua through Melanesia to New Guinea. The genus Howeavis (Mathews 1912, type rufifrons) brings up the end and applies to the rufous fantails, which range from Sulawesi out into the Pacific.
Four species are added to the Rhipiduridae (Fantails) based on Sánchez-González and Moyle (2011).
- Pied Fantail, Leucocirca javanica is split into Malaysian Pied-Fantail, Leucocirca javanica (including longicauda), and Philippine Pied-Fantail, Leucocirca nigritorquis.
- Blue Fantail, Cyanonympha superciliaris, is split into Visayan Blue-Fantail, Cyanonympha samarensis (including apo, which Sánchez-González and Moyle note is not genetically distinct), and Mindanao Blue-Fantail, Cyanonympha superciliaris.
- Tablas Fantail, Cyanonympha sauli, and Visayan Fantail, Cyanonympha albiventris, are split from Blue-headed Fantail, Cyanonympha cyaniceps. The race pinicola remains with cyaniceps. However, Sánchez-González and Moyle found evidence of a possible species-level split within cyaniceps on Luzon that does not match the usual division into pinicola and cyaniceps.
Sánchez-González and Moyle also found some evidence that may also indicate a species-level split within Black-and-cinnamon Fantail, Cyanonympha nigrocinnamomea. However, it may just reflect clinal changes within the species (as appears to be the case with plumage).
- Brown-capped Fantail, "Leucocirca1" diluta
- Sooty Thicket-Fantail, "Leucocirca2" threnothorax
- Black Thicket-Fantail, "Leucocirca2" maculipectus
- White-bellied Thicket-Fantail, "Leucocirca2" leucothorax
- Malaysian Pied-Fantail, Leucocirca javanica
- Philippine Pied-Fantail, Leucocirca nigritorquis
- Willie-wagtail, Leucocirca leucophrys
- White-browed Fantail, Leucocirca aureola
- White-spotted Fantail, Leucocirca albogularis
- White-throated Fantail, Leucocirca albicollis
- Black Fantail, "Rhipidura" atra
- Rufous-tailed Fantail, Neomyias phoenicurus
- White-bellied Fantail, Neomyias euryurus
- Spotted Fantail, Neomyias perlatus
- Cinnamon-tailed Fantail, Neomyias fuscorufus
- Northern Fantail, Neomyias rufiventris
- White-winged Fantail, Neomyias cockerelli
- Black-and-cinnamon Fantail, Cyanonympha nigrocinnamomea
- Visayan Blue-Fantail, Cyanonympha samarensis
- Mindanao Blue-Fantail, Cyanonympha superciliaris
- Blue-headed Fantail, Cyanonympha cyaniceps
- Tablas Fantail, Cyanonympha sauli
- Visayan Fantail, Cyanonympha albiventris
- Chestnut-bellied Fantail, Rhipidura hyperythra
- Friendly Fantail, Rhipidura albolimbata
- Samoan Fantail, Rhipidura nebulosa
- Kadavu Fantail, Rhipidura personata
- Streaked Fantail, Rhipidura verreauxi
- Brown Fantail, Rhipidura drownei
- Makira Fantail, Rhipidura tenebrosa
- Rennell Fantail, Rhipidura rennelliana
- Mangrove Fantail, Rhipidura phasiana
- Gray Fantail, Rhipidura albiscapa
- New Zealand Fantail, Rhipidura fuliginosa
- Rufous-backed Fantail, Howeavis rufidorsus
- Dimorphic Fantail, Howeavis brachyrhynchus
- Bismarck Fantail, Howeavis dahli
- Mussau Fantail, Howeavis matthiae
- Malaita Fantail, Howeavis malaitae
- Arafura Fantail, Howeavis dryas
- Pohnpei Fantail, Howeavis kubaryi
- Manus Fantail, Howeavis semirubrus
- Rufous Fantail, Howeavis rufifrons
- Palau Fantail, Howeavis lepidus
- Streak-breasted Fantail, Howeavis dedemi
- Tawny-backed Fantail, Howeavis superfluus
- Rusty-bellied Fantail, Howeavis teysmanni
- Long-tailed Fantail, Howeavis opistherythrus
Dicruridae: Drongos Vigors, 1825 (1824)
4 genera, 25 species HBW-14
 |
| Click for Drongo species tree |
|---|
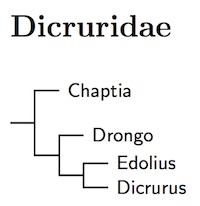
The ordering of the drongos is based on Pasquet et al. (2007) and Jønsson et al. (2016). The Bronzed Drongo has been put in its own genus Chaptia (Hodgson 1836). The White-bellied Drongo has also been placed in the monotypic genus Drongo (Tickell 1833). I've moved the western drongos into genus Edolius (Cuvier 1816, type forficatus). The eastern drongos remain in Dicrurus.
- Bronzed Drongo, Chaptia aenea
- White-bellied Drongo, Drongo caerulescens
- Square-tailed Drongo, Edolius ludwigii
- Shining Drongo, Edolius atripennis
- Ashy Drongo, Edolius leucophaeus
- Mayotte Drongo, Edolius waldenii
- Aldabra Drongo, Edolius aldabranus
- Crested Drongo, Edolius forficatus
- Grand Comoro Drongo, Edolius fuscipennis
- Velvet-mantled Drongo, Edolius modestus
- Fork-tailed Drongo, Edolius adsimilis
- Black Drongo, Edolius macrocercus
- Lesser Racket-tailed Drongo, Dicrurus remifer
- Greater Racket-tailed Drongo, Dicrurus paradiseus
- Crow-billed Drongo, Dicrurus annectans
- Sri Lanka Drongo, Dicrurus lophorinus
- Andaman Drongo, Dicrurus andamanensis
- Spangled Drongo, Dicrurus bracteatus
- Paradise Drongo, Dicrurus megarhynchus
- Hair-crested Drongo, Dicrurus hottentottus
- Sumatran Drongo, Dicrurus sumatranus
- Balicassiao, Dicrurus balicassius
- Tablas Drongo, Dicrurus menagei
- Sulawesi Drongo, Dicrurus montanus
- Wallacean Drongo, Dicrurus densus
Ifritidae: Ifrit Schodde and Christidis, 2014
1 genus, 1 species HBW-12, in Eupetidae (pg. 373)
The exact placement of Ifrita, Melampitta, and the Corcoracidae have been a problem. Aggerbeck et al. (2014) found them in a decently supported clade with the birds-of-paradise, but the exact relationship between them had less support.
An alternative view is that Ifrita is a monarch. Norman et al. (2009a) found Ifrita sister to a clade containing Monarcha and Myiagra. Jønsson et al. (2011b) has better taxon sampling and provides mixed support for considering it a basal monarch flycatcher. Jønsson et al. (2016) concur that Ifrita is sister to the monarchs, but with good support. However, the division between them is deep, deserving of family status. They place the monarchs and Ifrita sister to the bird-of-paradise clade. This is also consistent with Dumbacher et al. (2008) finding Ifrita related to Melampitta and the birds-of-paradise as those were the closest things to monarchs that were included in their analysis.
- Ifrit / Blue-capped Ifrit, Ifrita kowaldi
Paradisaeidae: Birds-of-paradise Vigors, 1825
16 genera, 43 species HBW-14
 |
| Click for Birds-of-paradise species tree |
|---|
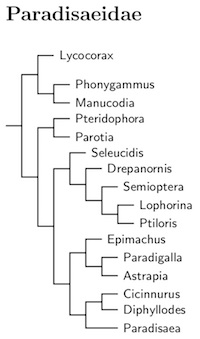
The phylogeny here follows Irestedt et al. (2009a) with corrections from Irestedt et al. (2017), allowing the riflebirds to be retained in genus Ptiloris.
There is a deep division between the manucodes (and paradise-crow) and the other birds-of-paradise (ca. 18 mya, Jønsson et al., 2016). This is reflected in the two subfamilies used here.
Based on Irestedt et al. (2017), the Superb Bird-of-paradise, Lophorina superba has been split into:
- Vogelkop Superb Bird-of-Paradise, Lophorina superba, inc. niedda.
- Greater Superb Bird-of-paradise, Lophorina latipennis, inc. addenda, feminina and latipennis
- Lesser Superb Bird-of-Paradise, Lophorina minor, monotypic
Note that Irestedt et al. argue that superba does not apply to birds from the Vogelkop Mountains, but to those usually called feminina. They establish a new name inopinata for the Vogelkop birds and refer to the Vogelkop Bird-of-paradise as Lophorina niedda. They also call the Greater Bird-of-paradise Lophorina superba. Given that the type of superba has been considered to be from the Vogelkop, I didn't find their arguments convincing.
Also, I've added Raggi's Bird-of-Paradise as the primary name of Paradisaea
raggiana, the point being that it is named for Francesco Raggi.
Phonygamminae: Manucodes G.R. Gray, 1846
- Paradise-crow, Lycocorax pyrrhopterus
- Trumpet Manucode, Phonygammus keraudrenii
- Glossy-mantled Manucode, Manucodia ater
- Jobi Manucode, Manucodia jobiensis
- Crinkle-collared Manucode, Manucodia chalybatus
- Curl-crested Manucode, Manucodia comrii
Paradisaeinae: Birds-of-paradise Vigors, 1825
- King-of-Saxony Bird-of-paradise, Pteridophora alberti
- Queen Carola's Parotia, Parotia carolae
- Bronze Parotia, Parotia berlepschi
- Western Parotia, Parotia sefilata
- Wahnes's Parotia, Parotia wahnesi
- Lawes's Parotia, Parotia lawesii
- Eastern Parotia, Parotia helenae
- Twelve-wired Bird-of-paradise, Seleucidis melanoleucus
- Black-billed Sicklebill, Drepanornis albertisi
- Pale-billed Sicklebill, Drepanornis bruijnii
- Standardwing, Semioptera wallacii
- Lesser Superb Bird-of-paradise, Lophorina minor
- Vogelkop Superb Bird-of-paradise, Lophorina superba
- Greater Superb Bird-of-paradise, Lophorina latipennis
- Victoria's Riflebird, Ptiloris victoriae
- Paradise Riflebird, Ptiloris paradiseus
- Magnificent Riflebird, Ptiloris magnificus
- Growling Riflebird, Ptiloris intercedens
- Black Sicklebill, Epimachus fastosus
- Brown Sicklebill, Epimachus meyeri
- Long-tailed Paradigalla, Paradigalla carunculata
- Short-tailed Paradigalla, Paradigalla brevicauda
- Arfak Astrapia, Astrapia nigra
- Splendid Astrapia, Astrapia splendidissima
- Huon Astrapia, Astrapia rothschildi
- Ribbon-tailed Astrapia, Astrapia mayeri
- Princess Stephanie's Astrapia, Astrapia stephaniae
- King Bird-of-paradise, Cicinnurus regius
- Wilson's Bird-of-paradise, Diphyllodes respublica
- Magnificent Bird-of-paradise, Diphyllodes magnificus
- Blue Bird-of-paradise, Paradisaea rudolphi
- Emperor Bird-of-paradise, Paradisaea guilielmi
- Red Bird-of-paradise, Paradisaea rubra
- Goldie's Bird-of-paradise, Paradisaea decora
- Lesser Bird-of-paradise, Paradisaea minor
- Greater Bird-of-paradise, Paradisaea apoda
- Raggi's Bird-of-paradise / Raggiana Bird-of-paradise, Paradisaea raggiana
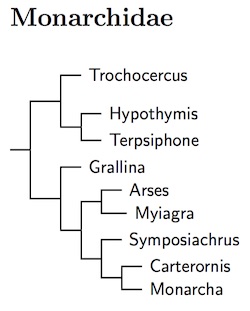
Monarchidae: Monarchs Bonaparte, 1854
10 genera, 102 species HBW-11
 |
| Click for Monarchidae species tree |
|---|
The current version of the Monarchidae is based on the comprehesive analysis by Andersen et al. (2015b), who included data from over 90% of the species recognized by TiF. They included multiple individuals from most species and a good selection of subspecies. This has been combined with the temporal analysis of Jønsson et al. (2016) to redraw the generic boundaries.
The genetics of the Monarchidae were previously studied by Pasquet et al. (2002), Filardi and Moyle (2005), and Filardi and Smith (2005). It was clear that the traditional Monarcha itself is paraphyletic. Christidis and Boles (2008) discussed appropriate generic names. TiF had adopted those, Monarcha into three genera: Carterornis, Monarcha, and Symposiachrus. Note that Metabolus has been absorbed into Monarcha based on Filardi and Moyle (2005) and Andersen et al. (2015b).
Andersen et al. (2015b) made it clear that additional genera would have to be named to keep several genera in Monarchinae. Jønsson et al. (2016) found that many of the involved species were closely related, closely enough that most would fit comfortably in a single genus (they suggested even more generic lumping). As a result, 5 genera have been merged into Monarcha. These are all part of the Monarcha radiation into the Pacific. Thus Chasiempis (Hawaii), Clytorhynchus (Fiji, Vanuatu, and the Solomons), Mayrornis (Fiji and the Solomons), Neolalage (Vanuatu), and Pomarea (Marquesas, Society, and Cook Islands) have been merged into Monarcha (which already included birds from the Bismarcks, Solomons, Carolines, Marianas, and Somoa, as well as Australia and New Guinea). This radiation appears to have occurred within the last 5-7 million years.
Historically, Pomarea pomarea has been used for both birds from Tahiti and Maupiti. Dickinson et al. (2019) argued that the Maupiti Monarch, Monarcha pomareus, Lesson and Garnot, 1828, should be restricted to the Tahitian birds, and that Monarcha maupitiensis, Garnot, 1829, should be used for the Maupiti birds. Finally, the Muscicapa nigra Sparrman, 1785 has priority over Muscicapa pomarea so the Tahitian birds are called Monarcha niger.
Fabre et al. (2012) has motivated some changes in species limits and the linear order within the genera. Their analysis found that the Pale-blue Monarch, Hypothymis puella, has been long-separated from the Black-naped Monarch, Hypothymis azurea. They only sampled three representatives of this complex, but HBW-11 suggests that puella should include puella, aeria, blasii and catarmanensis. Clements, which treats it as a separate species, only includes puella and blasii, leaving the other two subspecies in H. azurea.
The Fabre et al. analysis included 13 of the 15 recognized subspecies of the Asian Paradise-Flycatcher, Terpsiphone paradisi. They found three clades in the complex, two of which were more closely to other species than to the other clades. Interestingly enough, these three clades correspond to the three species recognized in the 19th century: Terpsiphone paradisi, Terpsiphone affinis, and Terpsiphone incei. A closer look shows that two of the subspecies are rather distantly related to the rest of the Terpsiphone affinis group, and I think it makes sense to recognize them as a separate species, Terpsiphone floris, giving us a total of four species.
Terpsiphone floris is visually distinct and seems to be separated from the affinis group by about 1.5 million years. This suggests it is probably best treated as a species. Its scientifc name creasted a minor problem as both subspecies appeared to have been named in the same year. Mees (2006, pg. 174) notes that floris was named either in late 1893 or the first few days of 1894. In contrast, sumbaensis was named in the January 1894 issue of the Journal für Ornithologie, which I think was running somewhat behind schedule (4-6 months in 1890). Thus floris seems to have priority. The 3 south Asian subspecies all have markedly longer crests. This fact and geography are why I've included burmae and saturatior in the affinis group.
The subspecies are allocated as follows:
- Amur Paradise-Flycatcher, Terpsiphone incei (monotypic);
- Indian Paradise-Flycatcher, Terpsiphone paradisi, with subspecies paradisi, leucogaster, and ceylonensis;
- Oriental Paradise-Flycatcher, Terpsiphone affinis, with subspecies nicobarica, burmae, indochinensis, affinis, procera, insularis, borneensis, and presumably burmae and saturatior;
- Lesser Sunda Paradise-Flycatcher, Terpsiphone floris, with two subspecies, sumbaensis and floris.
Fabre et al. (2012) also provides genetic support for separating the Luzon Paradise-Flycatcher, Terpsiphone unirufa, from the Rufous Paradise-Flycatcher, Terpsiphone cinnamomea. However, it provides no support for also separating T. c. talautensis as has sometimes been suggested. Finally, Fabre et al. found that the Rufous-vented Paradise-Flycatcher, Terpsiphone rufocinerea, is genetically close to African Paradise-Flycatcher, Terpsiphone viridis. These taxa also hybridize (HBW-11). The account in HBW-11 suggested that they may be conspecific, that genetic data should be consulted. Well, the current genetic data suggests it is most appropriate to treat them as a single species, African Paradise-Flycatcher. Fabre et al., also found evidence that there might be two species within the African Paradise-Flycatcher complex, but not rufocinerea. Further study is warranted.
The Monarcha branch splits into three main groups: Arses and Myiagra; Grallina; and Monarcha and allies. Fabre et al. (2014) have anaylized most of the Arses/Myiagra clade. Although Ochre-collared Monarch, Arses insularis, is basal in Arses, species boundaries for the rest of Arses may need adjustment. The taxonomic position of Grallina has been controversial, but Filardi and Moyle (2005) have resolved its position as sister to the Monarcha group.
As recommended by Andersen et al. (2015b), Chestnut-throated Flycatcher, Myiagra castaneigularis (inc. whitneyi), has been split from Azure-crested Flycatcher, Myiagra azureocapilla. In additiona, I have split White-cheeked Monarch, Symposiachrus malaitae, from Solomons Monarch, Symposiachrus barbatus.
Cibois et al. (2004) addressed the species status of the former Pomarea Monarchs of the central Pacific. VanderWerf (2007) showed that the forms of Elepaio on different islands were relatively unresponsive to each other's songs, while VanderWerf et al. (2010) shows they are as genetically distinct as the species in the former genus Pomarea. The combination of reasonable genetic distance and evidence of biological separation results in a split of the Elepaio, Monarcha sandwichensis, into 3 species: Kauai Elepaio, Monarcha sclateri, Oahu Elepaio, Monarcha ibidis, and Hawaii Elepaio, Monarcha sandwichensis.
Terpsiphoninae: Paradise-Flycatchers and alliesShelley, 1896 (1852)
- Blue-headed Crested-Flycatcher, Trochocercus nitens
- Blue-mantled Crested-Flycatcher, Trochocercus cyanomelas
- Black-naped Monarch, Hypothymis azurea
- Pale-blue Monarch, Hypothymis puella
- Short-crested Monarch, Hypothymis helenae
- Celestial Monarch, Hypothymis coelestis
- Luzon Paradise-Flycatcher, Terpsiphone unirufa
- Rufous Paradise-Flycatcher, Terpsiphone cinnamomea
- Blue Paradise-Flycatcher, Terpsiphone cyanescens
- Amur Paradise-Flycatcher, Terpsiphone incei
- Japanese Paradise-Flycatcher, Terpsiphone atrocaudata
- Oriental Paradise-Flycatcher, Terpsiphone affinis
- Lesser Sunda Paradise-Flycatcher, Terpsiphone floris
- Mascarene Paradise-Flycatcher, Terpsiphone bourbonnensis
- Indian Paradise-Flycatcher, Terpsiphone paradisi
- Sao Tome Paradise-Flycatcher, Terpsiphone atrochalybeia
- Malagasy Paradise-Flycatcher, Terpsiphone mutata
- Seychelles Paradise-Flycatcher, Terpsiphone corvina
- African Paradise-Flycatcher, Terpsiphone viridis
- Red-bellied Paradise-Flycatcher, Terpsiphone rufiventer
- Bedford's Paradise-Flycatcher, Terpsiphone bedfordi
- Bates's Paradise-Flycatcher, Terpsiphone batesi
- Annobon Paradise-Flycatcher, Terpsiphone smithii
Monarchinae: Monarch Flycatchers Bonaparte, 1854
- Magpie-lark, Grallina cyanoleuca
- Torrent-lark, Grallina bruijnii
- Pied Monarch, Arses kaupi
- Ochre-collared Monarch, Arses insularis
- Frilled Monarch, Arses telescopthalmus
- Frill-necked Monarch, Arses lorealis
- Shining Flycatcher, Myiagra alecto
- Velvet Flycatcher, Myiagra hebetior
- Chestnut-throated Flycatcher, Myiagra castaneigularis
- Azure-crested Flycatcher, Myiagra azureocapilla
- Palau Flycatcher, Myiagra erythrops
- Broad-billed Flycatcher, Myiagra ruficollis
- Paperbark Flycatcher, Myiagra nana
- Restless Flycatcher, Myiagra inquieta
- Oceanic Flycatcher, Myiagra oceanica
- Pohnpei Flycatcher, Myiagra pluto
- Samoan Flycatcher, Myiagra albiventris
- Melanesian Flycatcher, Myiagra caledonica
- Makira Flycatcher, Myiagra cervinicauda
- Vanikoro Flycatcher, Myiagra vanikorensis
- Steel-blue Flycatcher, Myiagra ferrocyanea
- Moluccan Flycatcher, Myiagra galeata
- Guam Flycatcher, Myiagra freycineti
- Biak Black Flycatcher, Myiagra atra
- Leaden Flycatcher, Myiagra rubecula
- Satin Flycatcher, Myiagra cyanoleuca
- Rufous Monarch, Symposiachrus rubiensis
- Black Monarch, Symposiachrus axillaris
- Hooded Monarch, Symposiachrus manadensis
- Mussau Monarch, Symposiachrus menckei
- Black-tailed Monarch, Symposiachrus verticalis
- White-tipped Monarch, Symposiachrus everetti
- Spectacled Monarch, Symposiachrus trivirgatus
- Spot-winged Monarch, Symposiachrus guttula
- Black-chinned Monarch, Symposiachrus boanensis
- White-tailed Monarch, Symposiachrus leucurus
- Manus Monarch, Symposiachrus infelix
- Black-bibbed Monarch, Symposiachrus mundus
- Black-tipped Monarch, Symposiachrus loricatus
- Kofiau Monarch, Symposiachrus julianae
- Biak Monarch, Symposiachrus brehmii
- Solomons Monarch, Symposiachrus barbatus
- White-cheeked Monarch, Symposiachrus malaitae
- Kolombangara Monarch, Symposiachrus browni
- White-collared Monarch, Symposiachrus vidua
- Flores Monarch, Symposiachrus sacerdotum
- Golden Monarch, Carterornis chrysomela
- White-eared Monarch, Carterornis leucotis
- White-naped Monarch, Carterornis pileatus
- Rarotonga Monarch, Monarcha dimidiatus
- Tinian Monarch, Monarcha takatsukasae
- Yap Monarch, Monarcha godeffroyi
- Tahiti Monarch, Monarcha niger
- Maupiti Monarch, Monarcha maupitiensis
- Chuuk Monarch, Monarcha rugensis
- Island Monarch, Monarcha cinerascens
- Black-faced Monarch, Monarcha melanopsis
- Black-winged Monarch, Monarcha frater
- Chestnut-bellied Monarch, Monarcha castaneiventris
- Bougainville Monarch, Monarcha erythrostictus
- White-capped Monarch, Monarcha richardsii
- Versicolored Monarch, Monarcha versicolor
- Iphis Monarch, Monarcha iphis
- Eiao Monarch, Monarcha fluxus
- Nuku Hiva Monarch, Monarcha nukuhivae
- Ua Pou Monarch, Monarcha mirus
- Marquesan Monarch, Monarcha mendozae
- Fatu Hiva Monarch, Monarcha whitneyi
- Kauai Elepaio, Monarcha sclateri
- Oahu Elepaio, Monarcha ibidis
- Hawaii Elepaio, Monarcha sandwichensis
- Buff-bellied Monarch, Monarcha banksianus
- Vanikoro Monarch, Monarcha schistaceus
- Slaty Monarch, Monarcha lessoni
- Rennell Shrikebill, Monarcha hamlini
- Black-throated Shrikebill, Monarcha nigrogularis
- Southern Shrikebill, Monarcha pachycephaloides
- Fiji Shrikebill, Monarcha vitiensis
- Santa Cruz Shrikebill, Monarcha sanctaecrucis
Corcoracidae: Australian Mudnesters Mathews, 1925
2 genera, 2 species HBW-14, as Struthideidae
The two mudnesters are a strongly supported family in Jønsson et al. (2016). Their closest relatives are the birds-of-paradise, from whom they are separated by around 22 million years of evolution.
The scientific name of the White-winged Chough is now Corcorax melanorhamphos, not melanoramphos as the ICZN has resolved the controversy over the name (Opinion 2380). In the same decision, they conserved the family name Corcoracidae over Struthideidae.
- White-winged Chough, Corcorax melanorhamphos
- Apostlebird, Struthidea cinerea
Melampittidae: Melampitta Schodde and Christidis, 2014
1 genus, 2 species HBW-12, in Eupetidae (pp. 372-3)
Reddy and Cracraft (2007) were the first to include both species of melampitta in the same analysis. They found them sister, and closest to the mudnesters. Jønsson et al. (2016) also included both melampittas and found strong support for their sister status,
Jønsson et al. (2011b) did not place the melampittas securely with either the mudnesters or birds-of-paradise. Their figure S1-A shows the latter while S1-B shows the former. Aggerbeck et al. (2014) and Jønsson et al. (2016) found them sister to the mudnesters plus the birds-of-paradise, and that is followed here.
- Lesser Melampitta, Melampitta lugubris
- Greater Melampitta, Melampitta gigantea
Laniidae: Shrikes Rafinesque, 1815
6 genera, 37 species HBW-13
The Crested Jay, Platylophus galericulatus, has been placed sister to the two White-crowned Shrikes (Eurocephalus). Jønsson et al. (2016) suggested they may be congeneric based on estimated divergence time, but there is a huge amount of uncertainty. It would be premature to merge the two genera at this time.
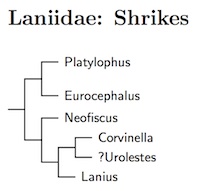 Jønsson et al. (2016) also found Lanius to be paraphyletic. As
a result, the Long-tailed Fiscal has been moved to the monotypic genus
Neofiscus (Roberts 1922) as Neofiscus cabanisi.
Jønsson et al. (2016) also found Lanius to be paraphyletic. As
a result, the Long-tailed Fiscal has been moved to the monotypic genus
Neofiscus (Roberts 1922) as Neofiscus cabanisi.
The genera Corvinella and probably Urolestes come next, followed by the remaining Lanius shrikes and fiscals. The arrangement of Lanius is rather conjectural and is inspired by a combination of Fuchs et al. (2011c), Gonzalez et al. (2008), Jønsson et al. (2016), Olsson et al. (2010), and Peer et al. (2011). Although I have created a tentative tree, it is too conjectural to include here.
The gray shrikes have been the subject of several genetic studies, including those of Mundy and Helbig (2004), Gonzalez et al. (2008), Klassert et al. (2008), and Olsson et al. (2010). Although Olsson et al.'s analysis is the most comprehensive, it still leaves a fair amount of uncertainty.
Mundy and Helbig (2004), analyzing DNA from several gray shrike taxa, found that the North American form invictus, conventionally considered part of excubitor, actually seemed to be more closely related to ludovicianus. Both Gonzalez et al. (2008) and Klassert et al. (2008) provided support for this, and added the subspecies meridionalis to the invictus-ludovicianus clade. This rendered the specis meridionalis polyphyletic. Olsson et al. (2010) analyzed many more of the gray shrike subspecies. Two of the fiscals, the Somali Fiscal and Taita Fiscal, seem to belong to the gray shrike group. The Taita Fiscal is most distant from the others, which divide into two clades as shown in the table below.
Exactly how to divide these groups into species is less than clear, and Olsson et al. suggest several possibilities. The one I've taken divides the first clade into 3 species, the monotypic Socotra Shrike, Lanius uncinatus (split from L. meridionalis); the Desert Shrike, Lanius elegans (also split from L. meridionalis); and the Great Gray Shrike, Lanius excubitor. The latter has gained the remainder of meridionalis (except meridionalis itself). I have also merged the Steppe Gray Shrike back into excubitor. The DNA differences between the various subspecies of Desert and Great Gray Shrike are large enough that they could represent more species, but small enough that all of them could be lumped into Great Gray Shrike. There is limited other evidence available. There are plumage differences, but one of the Desert Shrike subspecies is thought to interbreed some with one of the Great Gray Shrike subspecies.
The other clade includes six species. The Chinese Gray Shrike has been split into two monotypic species: Chinese Gray Shrike, Lanius sphenocercus, and Tibetan Shrike, Lanius giganteus. The two not only differ in plumage, but the DNA differences are consistent with species status. The Northern Shrike, Lanius borealis, includes not only North American subspecies, but also the eastern Palearctic subspecies of the former Northern/Great Gray complex. Finally, the Iberian Gray Shrike, which is closely related to the Northern Shrike, than to the rest of the Southern Gray Shirke complex, retains only one subspecies, meridionalis.
| The Gray Shrikes | ||
|---|---|---|
| Species | Subspecies | |
| Taita Fiscal, L. dorsalis | *dorsalis | |
| Socotra Shrike, L. uncinatus | *uncinatus | |
| Desert Shrike, L. elegans | *koenigi, *algeriensis, *elegans, *leucopygos | |
| Great Gray Shrike, L. excubitor | *excubitor, *homeyeri (inc. *leucopterus), *aucheri, theresae, *buryi, jebelmarrae, *pallidirostris, *lahtora | |
| Somali Fiscal, L. somalicus | *somalicus | |
| Loggerhead Shrike, L. ludovicianus | *excubitorides (inc. gambeli & sonoriensis), migrans, ludovicianus, miamensis, anthonyi, mearnsi, grinnelli, *mexicanus (inc. nelsoni) | |
| Chinese Gray Shrike, L. sphenocercus | *sphenocercus | |
| Tibetan Shrike, L. giganteus | *giganteus | |
| Northern Shrike, L. borealis | *borealis (inc. invictus), *sibiricus, *bianchii, *mollis, *funereus | |
| Iberian Gray Shrike, L. meridionalis | *meridionalis | |
| Subspecies based on Dickinson et al. (2003). *An asterisk indicates taxa sampled by Olsson et al. (2010). | ||
More recently, Fuchs et al. (2011c) have studied some of the fiscals. Their results suggest (1) the Uhehe Fiscal, Lanius marwitzi, is embedded in the Common Fiscal and should be merged (2) the Common Fiscal contains two species. The new alignment results in Northern Fiscal, Lanius humeralis (smithii, humeralis, capelli) and Southern Fiscal, Lanius collaris (aridicolus, pyrrhostictus, marwitzi, subcoronatus, collaris). Although the genetics appear to support this, it would be nice to see additional work backing it up, especially in the area where the two groups meet.
The Masked Shrike seems relatively and may belong with the gray shrikes, which is the smaller part of Lanius. The Gray-backed Fiscal may be basal among the remaining species. The Woodland/Lesser Gray Shrike seem to group together. There's a core fiscal group, tentatively mackinnoni through collaris, and a brown shirke group, tentatively including tigrinus through validirostris.
- Crested Jay, Platylophus galericulatus
- Northern White-crowned Shrike, Eurocephalus ruppelli
- Southern White-crowned Shrike, Eurocephalus anguitimens
- Long-tailed Fiscal, Neofiscus cabanisi
- Yellow-billed Shrike, Corvinella corvina
- Magpie Shrike, Urolestes melanoleucus
- Masked Shrike, Lanius nubicus
- Taita Fiscal, Lanius dorsalis
- Socotra Shrike, Lanius uncinatus
- Desert Shrike, Lanius elegans
- Great Gray Shrike, Lanius excubitor
- Somali Fiscal, Lanius somalicus
- Loggerhead Shrike, Lanius ludovicianus
- Northern Shrike, Lanius borealis
- Iberian Gray Shrike, Lanius meridionalis
- Chinese Gray Shrike, Lanius sphenocercus
- Tibetan Shrike, Lanius giganteus
- Gray-backed Fiscal, Lanius excubitoroides
- Woodchat Shrike, Lanius senator
- Lesser Gray Shrike, Lanius minor
- Mackinnon's Shrike, Lanius mackinnoni
- Sao Tome Fiscal, Lanius newtoni
- Northern Fiscal, Lanius humeralis
- Emin's Shrike, Lanius gubernator
- Souza's Shrike, Lanius souzae
- Southern Fiscal, Lanius collaris
- Tiger Shrike, Lanius tigrinus
- Bay-backed Shrike, Lanius vittatus
- Burmese Shrike, Lanius collurioides
- Isabelline Shrike, Lanius isabellinus
- Red-backed Shrike, Lanius collurio
- Red-tailed Shrike, Lanius phoenicuroides
- Bull-headed Shrike, Lanius bucephalus
- Long-tailed Shrike, Lanius schach
- Gray-backed Shrike, Lanius tephronotus
- Brown Shrike, Lanius cristatus
- Mountain Shrike, Lanius validirostris
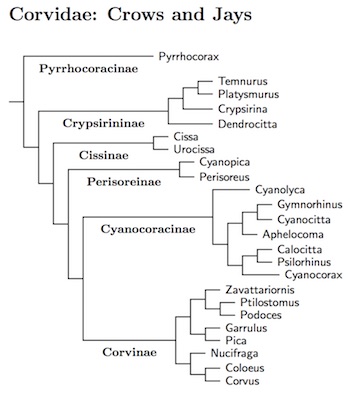
Corvidae: Crows, Jays Leach, 1820
24 genera, 133 species HBW-14
 |
| Click for Corvidae species tree |
|---|
The corvid genera are arranged based on Ericson et al. (2005), Jønsson et al. (2016), and, for the New World jays, Bonaccorso and Peterson (2007). I'm treating the natural division into 6 ancient clades as subfamilies. Jønsson et al. estimate these clades are all 17-20 million years old. A bit different arrangement is used by Ekman and Ericson (2006), but it includes fewer taxa.
Based on van Balen et al. (2013), the two allopatric forms of Short-tailed Green-Magpie are split into Bornean Green-Magpie, Cissa jefferyi, and Javan Green-Magpie, Cissa thalassina. Based on plumage, morphology, and voice, they argue that the differences between them are at a level typical of distinct species.
Although the Crested Jay, Platylophus galericulatus, has been considered a corvid, it is not. It is now in Laniidae.
The Iberian Magpie, Cyanopica cooki, has been split from Azure-winged Magpie, Cyanopica cyanus. These two widely-separated taxa show a significant amount of genetic differentiation. See Kryukov et al. (2004), Haring et al. (2007a), and Zhang et al. (2012).
I've split the Western Scrub-Jay into Woodhouse's Scrub-Jay, Aphelocoma woodhouseii, and California Scrub-Jay, Aphelocoma californica, based on Rice et al. (2003) and Delaney et al. (2008). This split failed to get the necessary 2/3's majority from AOU's NACC in 2009 (6-5 in favor). The sticking point involved questions about the hybrid zone. More recently, Gowen et al. (2014) studied the hybrid zone and argued in favor of the split. A further split of sumichrasti lost 4-7. I've also split the Mexican Jay into Transvolcanic Jay, Aphelocoma ultramarina, and Mexican Jay, Aphelocoma wollweberi, as in McCormack et al. (2008). It seems likely from their results that A. wollweberi will need at least one further split.
Bonaccorso (2009) was the primary source concerning the arrangement of Cyanolyca. The Calocitta to Cyanocorax clade is based on maximum parisomy and mitochondrial trees in Bonaccorso et al. (2010). This is consistent with their analysis of the full dataset, when applicable. I have retained Calocitta for the distinctive magpie-jays and usded Psilorhinus for the clade containing the Brown Jay. This is consistent with the maximum parsimony tree they found based on 5 genes. (The MP tree is described but not shown in the paper.)
Only mitochondrial DNA was used for the Azure Jay, Psilorhinus caeruleus. It ended up sister to the Brown Jay, but this was only weakly supported. Bonaccorso et al. describe the situation as a “virtual polytomy”. It is puzzling that the Azure Jay of extreme southern Brazil should be sister to the distinctive Brown Jay of Middle America, especially when there are similar jays right next door to the Azure Jay. I don't really trust this result. I should also note here that as part of the polytomy, Bonaccorso et al. found apparent paraphyly in the Violacenous Jay, with the Ecuadorian birds basal relative to those from Peru.
Since Bonaccorso et al. found substantial genetic distance between South American and Middle American green jays. These two groups are separated by Panama, Costa Rica, and Nicaragua; they look obviously different; vocalizations are different; there is substantial genetic distance. While they could be the same species, I think the balance of evidence favors splitting the 8 Middle American races as Green Jay, Cyanocorax luxuosus, and the South American races as Inca Jay, Cyanocorax yncas. This is how Hilty (2003) and Ridgely and Greenfield (2001) treat them, even though the AOU has not yet done so.
Campina Jay, described as Cyanocorax hafferi by Cohn-Haft et al. (2013), is treated as a subspecies of Azure-naped Jay, Cyanocorax heilprini (SACC #635).
Four of the Cyanocorax jays, melanocyaneus through beecheii are sometimes separated as the genus Cissilopha (Bonaparte, 1850). Although they form a clade, they are nested within Cyanocorax.
Previously, I had split Oriental Magpie, Pica bottanensis, including andersoni and serica has been split from Eurasian Magpie, Pica pica based on Lee et al. (2003), Kryukov et al. (2004), Haring et al. (2007a), and Zhang et al. (2012). Note that the inclusion of bottanensis is an educated guess, while that of andersoni and serica is based on genetics (Kryukov et al., 2004). The race camtschatica remains in Eurasian Magpie (Kryukov et al., 2004). The race asirensis is sometimes split as Arabian Magpie (e.g., HBW-14).
More recently, consideration of Kryukov et al. (2017) and the HBW-Checklist (del Hoyo and Collar, 2016) has resulted in further magpie splits. The Oriental Magpie, Pica bottanensis, has been split into Himalayan Magpie, Pica bottanensis (monotypic), and Oriental Magpie, Pica serica (including andersoni). Moreover, the Eurasian Magpie, Pica pica, has been split into Maghreb Magpie, Pica mauritanica (monotypic), Asir Magpie, Pica asirensis (monotypic), and Eurasian Magpie, Pica pica (all other subspecies). Note that genetic data is still lacking for asirensis and bottanensis.
Kryukov and Suzuki (2000) and Haring et al. (2007a) found that the jackdaws were rather distant from the other Corvus species. Accordingly, they are separated in the genus Coloeus, as recommended by Rasmussen and Anderton (2005). Although I had presumed that Collared Crow took the name Corvus torquatus because of this, thinking that otherwise Corvus monedula torquatus (Bechstein 1791) would have first claim on torquatus, this is apparently incorrect. Rather, Bechstein did not use the term to designate a species or subspecies. This did not make the name available as a species-group name, and so Lesson's use of Corvus torquatus for the Collared Crow in 1831 takes precendence over Corvus pectoralis for the Collared Crow. See BirdForum for more.
The arrangement within the genus Corvus is based on Jønsson et al. (2012a). This multi-gene analysis replaces a version based on the single-gene analysis of Haring et al. (2012). It has much better support for most of the nodes. A species tree for the crows, ravens, and jackdaws can be found by clicking the tree graphic to the right.
Jønsson et al. describe their results in terms of 8 clades. Clades II-VII are labeled on the tree (clade I is Coloeus). The clades don't include the Cape Crow, Corvus capensis, or Flores Crow, Corvus florensis, both of which are only weakly placed on the tree. The Flores Crow forms a strongly supported group with clades VII and VIII although the exact placement is somewhat uncertain. As for the Cape Crow, it is not a jackdaw, and seems to be relatively basal.
There are several areas needing further study. The Cuban and Hispaniolan races of Palm Crow seem to be separated by about 1 million years. This is consistent with them being separate species (as in IOC), or races of one species. The AOS now considers them separate species, Cuban Palm-Crow, Corvus minutus and Hispaniolan Palm-Crow, Corvus palmarum, primarily based on vocalizations.
The eastern and western Rooks are fairly distant relatives, and probably represent separate species (Haring et al. also found this). However, wider sampling is needed to clarify exactly where the boundary is, and whether there is a third rook species.
There is evidence that the Chihuahuan Raven, Corvus cryptoleucus, and possibly the Pied Crow, Corvus albus, are embedded in the Common Raven complex. Moreover, the Chihuahuan Raven seems more closely related to a mostly Californian clade of Common Ravens than either is to the other Common Ravens. See Feldman and Omland (2004) and Omland et al. (2000, 2006).
What seems to have happened is that the California population became separated from the other Common Ravens. During that time, the Chihuahuan Ravens separated from the California Common Ravens. Barriers to interbreeding developed between them, making Chihuahuan Raven a separate species. Later, the California Common Ravens came back into contact with the northern Common Ravens. However, they were still able to interbreed, and do so freely to this day (Webb et al., 2011), leaving only a mitochondrial signature of their divergent ancestry.
The Carrion and Hooded Crows are very closely related, and it is unclear whether they truly represent separate species. Further, Haring et al. (2012) found that some of the eastern individuals actually grouped with Collared Crow, Corvus torquatus (sometimes called pectoralis). In the same clade, the American Crow, Corvus brachyrhynchos and Northwestern Crow, Corvus caurinus are also quite closely related, and may not be separate species.
The Large-billed Crow complex has long been controversial (see the discussion in Dickinson et al., 2004). Martens et al. (2000) attempted to use acoustic evidence to resolve it. It remains confusing even with the new molecular evidence. Jønsson et al. (2012a) did not include either the nominate macrorhynchos or culminatus in their analysis. The did find that levaillantii and japonensis (including mandschuricus) where closer to each other than to kubaryi, and that all three grouped together to the exclusion of philippinus. Haring et al. sampled 25 individuals from this complex. They found evidence that colonorum belongs to the japonensis group, not with levaillantii. Their results also support species status for monotypic levaillantii, macrorhynchos, and philippinus, but did not clearly separate culminatus, japonensis, or the other subspecies. They also note incomplete data from some of these taxa. I'd like to see a closer study of this complex before attempting to restructure it. That said, I am tempted to split off philippinus.
Pyrrhocoracinae: Choughs G.R. Gray, 1846 (1831)
- Red-billed Chough, Pyrrhocorax pyrrhocorax
- Alpine Chough, Pyrrhocorax graculus
Crypsirininae: Treepies Swainson, 1831
- Ratchet-tailed Treepie, Temnurus temnurus
- Black Magpie, Platysmurus leucopterus
- Racket-tailed Treepie, Crypsirina temia
- Hooded Treepie, Crypsirina cucullata
- Rufous Treepie, Dendrocitta vagabunda
- Sumatran Treepie, Dendrocitta occipitalis
- Bornean Treepie, Dendrocitta cinerascens
- Gray Treepie, Dendrocitta formosae
- White-bellied Treepie, Dendrocitta leucogastra
- Collared Treepie, Dendrocitta frontalis
- Andaman Treepie, Dendrocitta bayleii
Cissinae: Green and Blue Magpies Kaup, 1855
- Common Green-Magpie, Cissa chinensis
- Indochinese Green-Magpie, Cissa hypoleuca
- Bornean Green-Magpie, Cissa jefferyi
- Javan Green-Magpie, Cissa thalassina
- Sri Lanka Blue-Magpie, Urocissa ornata
- Taiwan Blue-Magpie, Urocissa caerulea
- Yellow-billed Blue-Magpie, Urocissa flavirostris
- Red-billed Blue-Magpie, Urocissa erythroryncha
- White-winged Magpie, Urocissa whiteheadi
Perisoreinae: Northern Jays Informal?
- Azure-winged Magpie, Cyanopica cyanus
- Iberian Magpie, Cyanopica cooki
- Siberian Jay, Perisoreus infaustus
- Sichuan Jay, Perisoreus internigrans
- Gray Jay, Perisoreus canadensis
Cyanocoracinae: American Jays Kaup, 1855
- White-throated Jay, Cyanolyca mirabilis
- Dwarf Jay, Cyanolyca nanus
- Black-throated Jay, Cyanolyca pumilo
- Silvery-throated Jay, Cyanolyca argentigula
- Azure-hooded Jay, Cyanolyca cucullata
- Beautiful Jay, Cyanolyca pulchra
- Black-collared Jay, Cyanolyca armillata
- Turquoise Jay, Cyanolyca turcosa
- White-collared Jay, Cyanolyca viridicyanus
- Pinyon Jay, Gymnorhinus cyanocephalus
- Steller's Jay, Cyanocitta stelleri
- Blue Jay, Cyanocitta cristata
- Unicolored Jay, Aphelocoma unicolor
- Transvolcanic Jay, Aphelocoma ultramarina
- Mexican Jay, Aphelocoma wollweberi
- Florida Scrub-Jay, Aphelocoma coerulescens
- Woodhouse's Scrub-Jay, Aphelocoma woodhouseii
- California Scrub-Jay, Aphelocoma californica
- Island Scrub-Jay, Aphelocoma insularis
- Black-throated Magpie-Jay, Calocitta colliei
- White-throated Magpie-Jay, Calocitta formosa
- Brown Jay, Psilorhinus morio
- Azure Jay, Psilorhinus caeruleus
- Violaceous Jay, Psilorhinus violaceus
- Purplish Jay, Psilorhinus cyanomelas
- Curl-crested Jay, Psilorhinus cristatellus
- Green Jay, Cyanocorax luxuosus
- Inca Jay, Cyanocorax yncas
- White-tailed Jay, Cyanocorax mystacalis
- Bushy-crested Jay, Cyanocorax melanocyaneus
- Yucatan Jay, Cyanocorax yucatanicus
- San Blas Jay, Cyanocorax sanblasianus
- Purplish-backed Jay, Cyanocorax beecheii
- Tufted Jay, Cyanocorax dickeyi
- Black-chested Jay, Cyanocorax affinis
- Azure-naped Jay, Cyanocorax heilprini
- Cayenne Jay, Cyanocorax cayanus
- Plush-crested Jay, Cyanocorax chrysops
- White-naped Jay, Cyanocorax cyanopogon
Corvinae: Crows and Palearctic Jays Leach, 1820
- Stresemann's Bushcrow, Zavattariornis stresemanni
- Piapiac, Ptilostomus afer
- Henderson's Ground-Jay, Podoces hendersoni
- Biddulph's Ground-Jay, Podoces biddulphi
- Pander's Ground-Jay, Podoces panderi
- Pleske's Ground-Jay, Podoces pleskei
- Eurasian Jay, Garrulus glandarius
- Black-headed Jay, Garrulus lanceolatus
- Lidth's Jay, Garrulus lidthi
- Maghreb Magpie, Pica mauritanica
- Asir Magpie, Pica asirensis
- Himalayan Magpie, Pica bottanensis
- Oriental Magpie, Pica serica
- Eurasian Magpie, Pica pica
- Black-billed Magpie, Pica hudsonia
- Yellow-billed Magpie, Pica nuttalli
- Clark's Nutcracker, Nucifraga columbiana
- Spotted Nutcracker, Nucifraga caryocatactes
- Large-spotted Nutcracker, Nucifraga multipunctata
- Eurasian Jackdaw / Western Jackdaw, Coloeus monedula
- Daurian Jackdaw, Coloeus dauuricus
- Cuban Crow, Corvus nasicus
- White-necked Crow, Corvus leucognaphalus
- Jamaican Crow, Corvus jamaicensis
- Cuban Palm-Crow, Corvus minutus
- Hispaniolan Palm-Crow, Corvus palmarum
- Fish Crow, Corvus ossifragus
- Sinaloa Crow, Corvus sinaloae
- Tamaulipas Crow, Corvus imparatus
- Cape Crow, Corvus capensis
- Rook, Corvus frugilegus
- Hawaiian Crow, Corvus hawaiiensis
- Thick-billed Raven, Corvus crassirostris
- White-necked Raven, Corvus albicollis
- Common Raven / Northern Raven, Corvus corax
- Chihuahuan Raven, Corvus cryptoleucus
- Fan-tailed Raven, Corvus rhipidurus
- Brown-necked Raven, Corvus ruficollis
- Pied Crow, Corvus albus
- Somali Crow, Corvus edithae
- American Crow, Corvus brachyrhynchos
- Northwestern Crow, Corvus caurinus
- Collared Crow, Corvus torquatus
- Carrion Crow, Corvus corone
- Hooded Crow, Corvus cornix
- Flores Crow, Corvus florensis
- House Crow, Corvus splendens
- Mariana Crow, Corvus kubaryi
- Large-billed Crow, Corvus macrorhynchos
- Indian Jungle-Crow, Corvus culminatus
- Eastern Jungle-Crow, Corvus levaillantii
- Slender-billed Crow, Corvus enca
- Piping Crow, Corvus typicus
- Banggai Crow, Corvus unicolor
- Long-billed Crow, Corvus validus
- New Caledonian Crow, Corvus moneduloides
- Bougainville Crow, Corvus meeki
- White-billed Crow, Corvus woodfordi
- Violet Crow, Corvus violaceus
- Brown-headed Crow, Corvus fuscicapillus
- Gray Crow, Corvus tristis
- Little Crow, Corvus bennetti
- Bismarck Crow, Corvus insularis
- Torresian Crow, Corvus orru
- Australian Raven, Corvus coronoides
- Little Raven, Corvus mellori
- Forest Raven, Corvus tasmanicus