ARDEAE Wagler, 1830
Although the Jarvis et al. (2014) tree suggests that the Eurypgimorphae belong with the Aequornithes, support for this is a little soft there. The same relationship appears with 100% support in Stiller et al. (2024). It also appears in Jarvis et al. (2014), Prum et al. (2015), Suh et al. (2015), and Kimball et al. (2019).
One good reason to keep Eurypgimorphae and Aequornithes separate is the apparent ancientness of their separation. Kuhl et al. estimate the separation occurred very late in the Cretaceous period, at 66.2 mya. I think that's unlikely. Fortunately, their confidence interval extends into the Paleogene. Stiller et al. put a bit later date on the event, about 63 mya.
Sangster et al. (2022b) introduced the name Phaethoquornithes (Sangster, Braun, Johansson, Kimball, Mayr and Suh) for the clade I've been calling Ardae, largely because I can use it for a big clade containing the heron family Ardeidae that is not so dependent on the phylogeny. As Sangster et al. mention, their definition is based on the phylogeny in Kuhl et al. (2021) and Prum et al. (2015). It also works for Stiller et al. (2024), as well as the older papers by Jarvis et al. (2014), Suh et al. (2015), and Kimball et al. (2019).
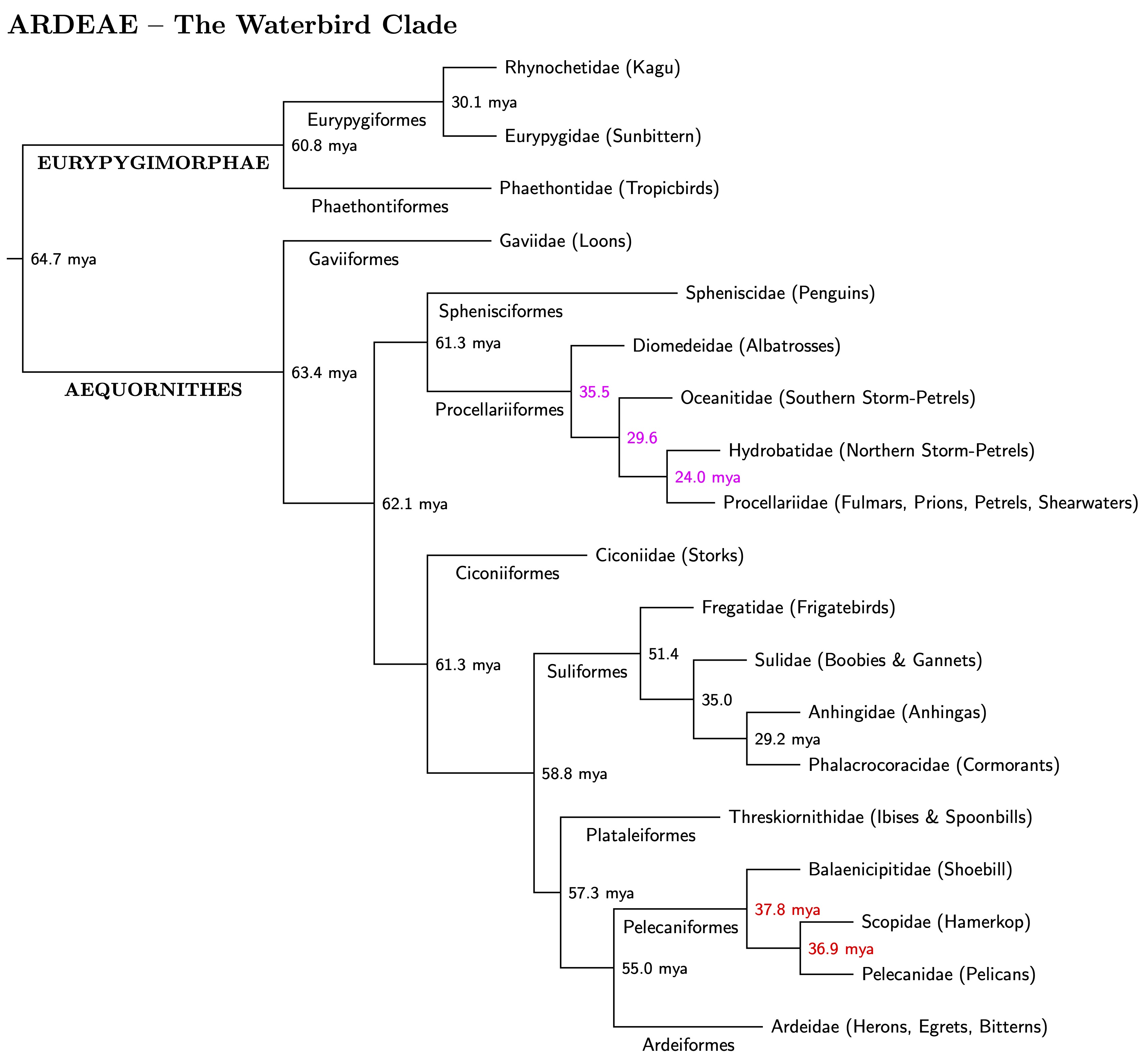 |
| The Waterbird Clade — Ardeae family tree |
|---|
The ages in black are consensus ages obtained by averaging those from Kuhl et al. (2021) and Stiller et al. (2024). The phylogenies are almost the same, with the only difference involving the three families in the Pelecaniformes. There are two problem areas, indicated by magenta and red ages.
The ages Stiller et al. found for the Procellariiformes (seabirds) seemed especially old to me. They are awfully similar for such old splits. Digging in the supplements to Stiller et al., I found they also seemed too old based on the fossils used for calibration (less than 2% probability according to distribution calculated for the supplement). If you haven't a skewed t distribution before, they were introduced by my former colleague Bruce Hansen (1994), back when we were both at the U. of Rochester. I decided to use the Kuhl et al. ages (the magenta ages). They are not far below the median of the distribution calculated by Stiller et al.
The red ages in the Pelecaniformes are warnings that the Kuhl et al. and Stiller et al. phylogenies differ slightly here. I used the Stiller et al. phylogeny and their ages. The Kuhl et al. dates are similar, but their phylogeny is (Pelicans, (Hamerkop, Shoebill)).
EURYPYGIMORPHAE Fürbringer, 1888
Traditional classifications of the tropicbirds have usually focused on the totipalmate feet and grouped them with the traditional Pelecaniformes because the Pelicaniformes also have totipalmate feet. Just think of the frigatebirds, boobies, anhingas, cormorants, and pelicans. The tropicbirds have long been recognized as being somewhat different. More recently, the pelicans and frigatebirds have also been considered a bit different. Morphological analyses continued to show this even in the phylogenetic era.
Cracraft (1985) considered the tropicbirds different enough that he divided the Pelicaniformes into two suborders: Phaethontes (tropicbirds) and Steganopodes (the rest). He further separated the frigatebirds as a infraorder and pelicans as a superfamily, leaving the boobies, cormorants, and anhingas more closely grouped. There is a lot of truth to this sort of arrangement.
However, what all thiis misses is that the totipalmate birds are not a natural group! Hedges and Sibley (1994) used DNA hybridization to argue that not only did the pelicans and frigatebirds not belong with the group, but that the tropicbird also didn't belong.
In fact, their Figure 2 is quite interesting. If you try to map it onto the tree I'm using, you find that the tropicbirds are outside the Aequornithes entirely. More recent DNA analyses based on sequences usually put the frigatebirds (but not pelicans) in a group with the boobies, gannets, cormorants, and darters. We follow that here.
More recent genetic studies, such as that of van Tuinen et al. (2001) have more decisively broken up the totipalmate group. In particular van Tuinen et al. correctly placed the pelicans close to the Hamerkop and Shoebill. The various studies supporting Metaves, including Fain and Houde (2004), Ericson et al. (2006a), and Hackett et al. (2008), all included the tropicbirds in Metaves, well separated from the rest of the totipalmate birds. They also all supported a close relationship between the Hamerkop, Shoebill, and pelicans.
Nonetheless, there were still problems with the tropicbirds. Using complete mitochondrial genomes, Gibb et al. (2013) found the tropicbirds nowhere near the rest of the “Pelicaniformes”. McCormack et al. (2013) placed the topicbirds near the Kagu in a grouping reminiscent of Metaves (but different). Finally, the data-intensive analysis by Jarvis et al. (2014) found that Eurypygiformes and Phaethontiformes were sister orders, with 100% bootstrap support. This is even more true of Kuhl et al. (2021) and Stiller et al. (2024). Moreover, they all placed the Kagu/Sunbittern/tropicbird group sister to Aequornithes, as is done here.
The orders Eurypygiformes and Phaethontiformes separated at 62.2 mya according to Kuhl et al., and at about 59 mya according to Stiller et al. That, and the obvious differencess between them, justifies their status as orders.
EURYPYGIFORMES Fürbringer, 1888
These two monotypic families (and orders!) already formed a strongly supported clade in Hackett et al. (2008). That is still true with Kuhl et al. (2021) and Stiller et al. (2024). Their affinities have long been unclear. They had recently been grouped near the cranes, but that appears incorrect. Ericson et al. (2006a) put them in Metaves near Columbea while Hackett et al. (2008) had them sister to the Strisores (also in Metaves). However, Jarvis et al. (2014) have them sister to the tropicbirds and near the waterbird group, Aequornithes.
Rhynochetidae: Kagu Carus, 1868
1 genus, 1 species HBW-3
- Kagu, Rhynochetos jubatus
Eurypygidae: Sunbittern Selby, 1840
1 genus, 1 species HBW-3
- Sunbittern, Eurypyga helias
PHAETHONTIFORMES Sharpe, 1891
Phaethontidae: Tropicbirds Brandt, 1840
1 genus, 3 species HBW-1
- Red-billed Tropicbird, Phaethon aethereus
- Red-tailed Tropicbird, Phaethon rubricauda
- White-tailed Tropicbird, Phaethon lepturus
AEQUORNITHES Mayr, 2011
The remainder of the Ardeae are in the waterbird clade, Aequornithes. Gibb et al. (2013) use the term “water-carnivore” to describe the birds in this clade. The Aequornithes include 8 closely related orders: Gaviiformes (loons), Sphenisciformes (penguins), Procellariiformes (petrels and shearwaters), Ciconiiformes (storks), Suliformes (frigatebirds, boobies, cormorants, darters), Plataleiformes (ibis), Pelecaniformes (shoebill, hamerkop, pelicans), and Ardeiformes (herons).
There is a lot of support for grouping these birds together (e.g., Cracraft et al, 2004; Ericson et al., 2006a; Gibb et al., 2007, 2013; Hackett et al, 2008; Morgan-Richards et al, 2008), Jarvis et al. (2014).
GAVIIFORMES Coues 1903
The Gaviiformes (Loons) are the oldest extant branch in the Aequornithes tree, dating back to the early Palaeocene. The oldest Gaviiforme fossil (Nasidytes ypresianus) is somewhat younger than that, dating back to about 54 mya (Mayr and Kitchener, 2022). Since the penguins are the second branch of Aequornithes, and the oldest fossil penguins are over 60 million years old, the Gaviiformes must be at least as old, hence early Paleocene.
The name Gaviiformes has been attributed to Wetmore & Miller, 1926, but Coues had already used Gaviae as a suborder in the second edition (1903) of his “Key to North American Birds” (pg. 1047). The term Colymbiformes has also been used. However, the ICZN eventually suppressed the genus Colymbus due to confusion about whether it applied to loons or grebes. Wikipedia has information on the ICZN decision resolving the controversy on it's Loon page.
Because of the ICZN decision, I have not tried to attribute priority of the name Gaviiformes to some earlier name of the order based on Colymbus, even though some cases may be unambiguous.
Loons and grebes may seem a strange combo, but until the DNA revolution, loons and grebes were generally considered relatives. That wasn't a crazy idea. Neither had any obvious relatives. Moreover, both are foot-propelled divers with the back legs positioned so far back that they can't walk on land.
So what does genetics have to say about loon taxonomy? In his MA thesis, Sprengelmeyer (2014) found that the Red-throated Loon is very distantly separated from the other loons. His point estimate of the age of the most recent common ancestor is 21.4 million years ago. In contrast, the most recent common ancestor for the other four loons was only estimated at 8.2 million years ago. It's reasonable to put the other loons in the same genus (Gavia), but the Red-throated Loon is really different.
Because it is really different, the Red-throated Loon not only gets its own genus, but its own subfamily. Unfortunately, there are no available names for this genus, and I will refer to it as "Gavia" until there is one.

Gaviidae: Loons J.A. Allen, 1897 (1840)
"Gaviinae": Red-throated Loon Informal
- Red-throated Loon, "Gavia" stellata
Gaviinae: Large Loons J.A. Allen, 1897 (1840)
- Arctic Loon / Black-throated Loon, Gavia arctica
- Pacific Loon, Gavia pacifica
- Common Loon, Gavia immer
- Yellow-billed Loon, Gavia adamsii
SPHENISCIFORMES Huxley, 1867
Spheniscidae: Penguins Bonaparte, 1831
6 genera, 19 species HBW-1
The penguin taxonomy follows Baker et al. (2006). Ksepka and Thomas (2012) add some morphological data. They obtained an almost identical topology, differing only in the branching order within Eudyptes.
Although the members of the pairs Macaroni/Royal and Snares/Fiordland are considered separate biological species, the pair Little/White-flippered are not. Christidis and Boles (2008) opined that it was premature to split them, and subsequent analysis have proven them correct. The complicated situation of the Little Penguin is analyzed in detail by Puecker et al. (2009), and I suspect it is not the last word on this. They found two clades, as did previous workers. However, they sampled many more penguins and found the clades did not divide as expected. In particular, there is no support for treating the White-flippered Penguin, Eudyptula minor albosignata as a separate species. Rather, there is a mostly Australian clade (with some New Zealand birds mostly from Otago and Omaru), and a clade covering the rest of New Zealand. Although most of the birds at Omaru seem to group with the Australian E. m. novaehollandiae, not all do. It appears likely that the type of E. m. minor, which is from Dusky Sound, belongs to the New Zealand clade. The significance of the presence of Australian clade birds at Otago/Omaru is yet to be fully understood. E.g., is there interbreeding? If so, how much? Although some uncertainty remains, it looks like two species are involved. The name Little Penguin has been official in Australia for some time, while Blue Penguin has been used in New Zealand, so it makes sense to call them Little Penguin, Eudyptula novaehollandiae, and Blue Penguin, Eudyptula minor.
More recently, Grosser et al. (2015), studying DNA from modern penguins, found that the Austalian lineage penguins in New Zealand were recent arrivals, probably within the last 1500 years. Then Grosser et al. (2016) sampled bones from 146 prehistoric penguins found in New Zealand. The bones prior to 1500 AD all came from Blue Penguins. The Australian lineage Little Penguins did not show up until sometime after 1500. They speculate that a population decline of the native Blue Penguins following the arrival of humans created an opening for Little Penguins to colonize the island.
The Macaroni/Royal and Snares/Fiordland pairs breed on different islands. The differences in appearance and DNA to are sufficient to allow treatment as separate species. In fact, the DNA difference seems to be less than between the Eudyptula clades (Baker et al., 2006), but the Eudyptula plumage differences are smaller and the situation on the breeding grounds is unclear.
Jouventin et al. (2006) make a good case for splitting Rockhopper Penguin into two biological species. I did not find the case for a three-way split compelling (Banks et al, 2006).
- King Penguin, Aptenodytes patagonicus
Click for Penguin tree - Emperor Penguin, Aptenodytes forsteri
- Adelie Penguin, Pygoscelis adeliae
- Gentoo Penguin, Pygoscelis papua
- Chinstrap Penguin, Pygoscelis antarcticus
- Little Penguin, Eudyptula novaehollandiae
- Blue Penguin, Eudyptula minor
- Humboldt Penguin, Spheniscus humboldti
- Galapagos Penguin, Spheniscus mendiculus
- African Penguin / Jackass Penguin, Spheniscus demersus
- Magellanic Penguin, Spheniscus magellanicus
- Yellow-eyed Penguin, Megadyptes antipodes
- Erect-crested Penguin, Eudyptes sclateri
- Royal Penguin, Eudyptes schlegeli
- Macaroni Penguin, Eudyptes chrysolophus
- Fiordland Penguin, Eudyptes pachyrhynchus
- Snares Penguin, Eudyptes robustus
- Rockhopper Penguin / Southern Rockhopper Penguin, Eudyptes chrysocome
- Tristan Penguin / Northern Rockhopper Penguin, Eudyptes moseleyi
PROCELLARIIFORMES Fürbringer, 1888
The arrangement of the Procellariiforme families follows Prum et al. (2015). A number of sources have been consulted concerning the species sequence. Austin (1996), Austin et al. (2004), Kennedy and Page (2002) and Penhallurick and Wink (2004) were generally useful in organizing the Procellariiformes. Concerning the latter, the comments by Rheindt and Austin (2005) should be noted. Prum et al. (2015), Welch et al. (2014), and Gangloff et al. (2012) were helpful concerning the Procellariidae.
Diomedeidae: Albatrosses G.R. Gray, 1840
4 genera, 21 species HBW-1
Traditionally, the 24 recognized albatross taxa have been grouped into 13 species.
Traditional Albatross Species Limits
24 taxa, 13 species
- Laysan Albatross, Phoebastria immutabilis
- Black-footed Albatross, Phoebastria nigripes
- Waved Albatross, Phoebastria irrorata
- Short-tailed Albatross, Phoebastria albatrus
- Royal Albatross, Diomedea epomophora
- Diomedea epomophora sanfordi
- Diomedea epomophora epomophora
- Wandering Albatross, Diomedea exulans
- Diomedea exulans dabbenena
- Diomedea exulans amsterdamensis
- Diomedea exulans antipodensis
- Diomedea exulans gibsoni
- Diomedea exulans exulans
- Sooty Albatross, Phoebetria fusca
- Light-mantled Albatross, Phoebetria palpebrata
- Yellow-nosed Albatross, Thalassarche chlororhynchos
- Thalassarche chlororhynchos chlororhynchos
- Thalassarche chlororhynchos carteri
- Grey-headed Albatross, Thalassarche chrysostoma
- Black-browed Albatross, Thalassarche melanophris
- Thalassarche melanophris melanophris
- Thalassarche melanophris impavida
- Buller's Albatross, Thalassarche bulleri
- Thalassarche bulleri bulleri
- Thalassarche bulleri platei
- Shy Albatross, Thalassarche cauta
- Thalassarche cauta cauta
- Thalassarche cauta steadi
- Thalassarche cauta eremita
- Thalassarche cauta salvini
Robertson and Nunn (1998) suggested a radical new taxonomy for albatrosses, elevating all 24 taxa to species level. This has caused a certain amount of controversy, and has not been universally accepted (e.g., Penhallurick and Wink, 2004; Penhallurick, 2012). The 4th edition of the highly regarded Howard and Moore checklist (Dickinson and Remsen, 2013) also continues to follow traditional albatross taxonomy. Their version is shown above.
Nonetheless, many other sources have moved toward the Robertson and Nunn taxonomy, and the TiF list uses a 21 species version. IOC 3.3 uses the same 21 species list as TiF. BirdLife International (ver. 5) additionally splits T. cauta and T.steadi for 22 species. The AOU's SACC has adopted the 3-way split of Thalassarche cauta used here (see proposals #155 and #255. Clements 6.7 accepts only this split and uses a 15 species list. The SACC also considered splitting Diomedea exulans into 4 species (see proposal #388). This was unable to gain the required 2/3's majority (the vote was 6-4 in favor of the split). Penhallurick (2012) makes a case for retaining the traditional classification.
How to treat slightly differentiated allopatric taxa, where the breeding ranges do not overlap, is often a thorny issue. If you read SACC proposal #388, you will see just how contentious it is.
My take on it is that there is some evidence of restricted gene flow between many of these taxa—a sign of legitimate biological species. Although the evidence is a long way from being convincing, I think it is enough to barely tip the scale in favor of the 21-species treatment below, at least for the present.
Burg and Croxall (2004), Bried et al. (2007), and Rains et al. (2011) provided support for most of the Robertson and Nunn splits in the Wandering Albatross group (except for D. antipodensis gibsoni), while Burg and Croxall (2001) examined the Black-browed/Gray-headed Albatross group. The Shy Albatrosses were studied by Abbott and Double (2003a, b). Interestingly, the Wandering Albatross in the narrow sense remains widespread even after the other taxa in the group (Tristan, Antipodes, and Amsterdam Albatrosses) are split off.
The phylogeny used here is based on Nunn and Stanley (1998) and Chambers et al. (2009). Finally, the term platei is often used for the northern populations of Buller's Albatross. It is said to refer instead to a juvenile of the southern population, in which case a new name is needed for the northern population (e.g., Chambers et al., 2009).
- Waved Albatross, Phoebastria irrorata

Click for Albatross tree - Short-tailed Albatross, Phoebastria albatrus
- Laysan Albatross, Phoebastria immutabilis
- Black-footed Albatross, Phoebastria nigripes
- Northern Royal Albatross, Diomedea sanfordi
- Southern Royal Albatross, Diomedea epomophora
- Tristan Albatross, Diomedea dabbenena
- Antipodean Albatross, Diomedea antipodensis
- Amsterdam Albatross, Diomedea amsterdamensis
- Wandering Albatross, Diomedea exulans
- Sooty Albatross, Phoebetria fusca
- Light-mantled Albatross, Phoebetria palpebrata
- Atlantic Yellow-nosed Albatross, Thalassarche chlororhynchos
- Indian Yellow-nosed Albatross, Thalassarche carteri
- Gray-headed Albatross, Thalassarche chrysostoma
- Black-browed Albatross, Thalassarche melanophris
- Campbell Albatross, Thalassarche impavida
- Buller's Albatross, Thalassarche bulleri
- White-capped Albatross / Shy Albatross, Thalassarche cauta
- Chatham Albatross, Thalassarche eremita
- Salvin's Albatross, Thalassarche salvini
Oceanitidae: Southern Storm-Petrels Forbes, 1882
5 genera, 9 species Not HBW Family
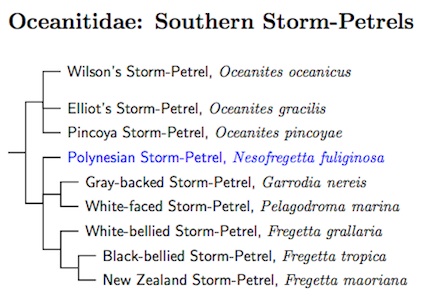
The split of the Storm-Petrels into two families was suggested by Nunn and Stanley (1998). See also Hackett et al. (2008) and Prum et al. (2015).
I've added the Pincoya Storm-Petrel, Oceanites pincoyae, described by Harrison et al. (2013).
I've also added the New Zealand Storm-Petrel, which was rediscovered in 2003 (Gaskin and Baird, 2005; Stephenson et al, 2008a). Details of the capture of one are on the Pterodroma Pelagics web site. Some uncertainty remained as to its identity after the initial reports, but a comparison with museum specimens (Stephenson et al, 2008b) removed any doubt that it was a New Zealand Storm-Petrel. A recent genetic analysis by Robertson et al. (2011), based partly on data from Nunn and Stanley (1998), showed that it belongs in genus Fregetta rather than the monotypic Pealeornis.
Robertson et al. (2011) also found that the Fregetta race leucogaster, often considered a subspecies of the White-bellied Storm-Petrel, is actually much more closely related to the Black-bellied Storm-Petrel. Whether it is a subspecies or distinct species is unclear at this point.
- Wilson's Storm-Petrel, Oceanites oceanicus
- Elliot's Storm-Petrel, Oceanites gracilis
- Pincoya Storm-Petrel, Oceanites pincoyae
- Polynesian Storm-Petrel, Nesofregetta fuliginosa
- Gray-backed Storm-Petrel, Garrodia nereis
- White-faced Storm-Petrel, Pelagodroma marina
- White-bellied Storm-Petrel, Fregetta grallaria
- Black-bellied Storm-Petrel, Fregetta tropica
- New Zealand Storm-Petrel, Fregetta maoriana
Hydrobatidae: Northern Storm-Petrels Mathews, 1912-13 (1865)
4 genera, 17 species HBW-1
The Hydrobatidae have been rearranged based on Nunn and Stanley (1998), Penhallurick and Wink (2004), and Sausner et al. (2016). This entails moving the Fork-tailed Storm-Petrel, Oceanodroma furcata, to the genus Hydrobates. Since O. furcata is the type species of Oceanodroma, and Hydrobates (Boie, 1822) has priority over Oceanodroma (Reichenbach, 1852), the name Oceanodroma must be given up.
One option would be to put the entire family in Hydrobates, as in H&M-4. I find it more helpful to treat each of the four clades as a separate genus. Fortunately, the supply of available names is more than adequate. Those that are relevant are Cymochorea (Coues 1864, type leucorhoa) and Halocyptena (Coues 1864, type microsoma), and Thalobata (Mathews and Hallstrom 1943, type castro).
I've grouped melania and matsudairae together as they are sometimes considered conspecific. That pair is sister to the microsoma/tethys pair, and all join Halocyptena. I've also grouped two other possibly conspecific pairs, tristrami and markhami, and monorhis and leucorhoa. Sausner et al. (2016) found that homochroa is close to leucorhoa, and that hornbyi is basal in this group. All of these go in Cymochorea.
That brings us to the basal group, the contentious Band-rumped (Madeiran) Storm-Petrel, Thalobata castro. Traditionally, it has been thought almost undifferentiated across the Atlantic and Pacific. Now we find that the genes reveal both substantial geographic and seasonal structure, enough that some recommend dividing it into a number of species (see Bolton (2007); Bolton et al., 2008; Friesen et al., 2007; Smith and Friesen, 2007; Smith et al., 2007).
In several locations, Band-rumped Storm-Petrel breeds in both the hot and cool seasons. Recent studies have found that the hot-season population is different from the cool-season population (e.g., Bolton, 2007; Frisen et al., 2007). The following table shows the island groups where Band-rumped Storm-Petrels breed, the season they breed, and applicable subspecific names. There may also be a population breeding on or near Sao Tome, but breeding sites have never been located. Further, it is unknown how closely the St. Helena and Ascension birds are related.
The breeding locations and seasons are:
| Location | Season | Subspecies | TiF Species |
|---|---|---|---|
| Ascension & St. Helena Islands | hot | helena | castro |
| Azores | hot | monteiroi | monteiroi |
| Azores Madeira Canaries Berlengas |
cool both cool cool |
castro | castro |
| Cape Verde Islands | protracted (cool) | jabejabe | jabejabe |
| Galapagos Islands | both | bangsi | cryptoleucura |
| Hawaiian Islands | hot | cryptoleucura | cryptoleucura |
| Japan | hot | kumagai | cryptoleucura |
Because they breed in the same location, there is a tendency to think of these as sympatric populations. Since they don't interbreed, they must be distinct species. QED.
Some have even suggested that castro be restricted to the birds breeding in Madiera (Desertas and Selvagem) during the hot season. The rest would be separated as Grant's Storm-Petrel, which does not yet have a scientific name. I find this hard to swallow. Based on Friesen et al. (2007) and Smith et al. (2007), the genetic distances appear to be quite small. Any separation between them is quite recent, perhaps within the Holocene.
Although Friesen et al. (2007) suggest the ancestral birds bred in the hot season, I don't really see this. The Cape Verde population is sister to the others and has a prolonged breeding season. If the ancestral population spread from there, one could easily see it adapting to local conditions that variously support breeding in the hot and/or cool seasons.
This suggests that considering them as sympatric gives the wrong impression. Rather, these populations occupy different niches that in some cases are separated temporally rather than geographically. They are better regarded as being adjacent (or even isolated) rather than overlapping.
This changes the picture. If we think of these populations as potential allospecies, they may not make the grade. There's not much differentiation. More evidence is needed, and there is more for some populations. Bolton (2007) used tape playback to explore whether there are pre-mating barriers to interbreeding. He investigated populations on the Cape Verde, Galapagos, and Azores islands. Although birds responded to calls of birds from their own islands, response to birds from other islands was weak and often no more than to unrelated control species.
This suggests that at least the subspecies tested — jabejabe (Cape Verde), bangsi (Galapagos), monteiroi (Azores hot season), and castro (Azores cold season only) — are distinct biological species. What about the other populations? We first consider the remaining Atlantic populations. Table 3 in Smith et al. (2007) addresses this issue. It shows that the northern Atlantic populations other than monteiroi are quite closely related (estimated divergence times from 100(!) to 17,000 years). Accordingly, I keep them all in T. castro. It also suggests that the birds from Ascension (and St. Helena?) are fairly close to the main populations of castro (divergence time 15,000-30,000 years, as opposed to about 100,000 years between monteiroi and castro, and 200,000-300,000 between jabejabe and either castro or monteiroi). Accordingly, I also treat helena as a form of T. castro.
 |
| Click for Storm-Petrel tree |
|---|
That brings us to the Pacific populations. We start with the hot and cool season breeders at the Galapagos Islands. Bolton (2007) found they did not respond to the calls of band-rumped storm-petrels from the Atlantic. Moreover, Smith et al. (2007) found divergence times of over 200,000 years between them and the Atlantic breeders. Finally, Smith and Friesen (2007) found only weak evidence that these involve a cryptic species, and suggested they are only as distinct from each other as subspecies. Here they are treated as part of the same species, distinct from the Atlantic species. The analysis of Freisen et al. (2007) found that the Japanese and Galapagos breeders form a separate clade. Returning to Table 3 of Smith et al. (2007), we also see that the Hawaiian breeders belong in this group. Moreover, the divergence time of 150,000-200,000 years does not compel us to treat them as separate species from each other in the absence of further evidence. Accordingly, I treat the Pacific populations of band-rumped storm-petrels as a single species, T. cryptoleucura, including bangsi (Galapagos) and kumagai (Japan).
When all is said and done, I treat the band-rumped storm-petrels as 4 species. These species are separated not only by breeding location, but by whether they breed in the hot or cool season. In some cases there is little genetic differentiation between hot or cool season breeders, or across islands. When there is no other evidence they form separate species, those populations are lumped together, either as T. castro or T. cryptoleucura.
The Leach's complex has also come under increased scrutiny. Townsend's Storm-Petrel, Cymochorea socorroensis, and Ainley's Storm-Petrel, Cymochorea cheimomnestes, have been split from Leach's Storm-Petrel, Cymochorea leucorhoa, based on a combination of Ainley (1980), Howell (2012), Adams et al. (2016), and the discussion in the proposal that was adopted in the 57th supplement to the AOU checklist.
Howell (2012) has raised the issue of whether chapmani should be treated as a separate species. It's generally considered that it's part of a cline with other Leach's Storm-Petrels on the Pacific coast. There's also a question of whether the Pacific Leach's are conspecific with the Atlantic Leach's. The name leucorhoa has a type locality off Picardy (France), and so applies to the Atlantic birds, while beali (type locality Sitka, Alaska) has very narrow priority for the Pacific Leach's, even if chapmani is included. Priority! (Dickinson et al., 2011) dates beali to March 20, 1906 (second 1906 issue of Condor) and chapmani to April 5, 1906 (second 1906 issue of Auk).
Leach's calls are apparently quite similar between the Atlantic and Pacific. Bicknell et al. (2012) found a small continuing gene flow from the Pacific to Atlantic (but not vice-versa) and estimated the main divergence between them at about 13,000 years ago, which does not suggest species status. To me, the amount of haplotype diversity is surprising for such a small difference (see Figures 1 and 3), and I suspect that the two have been substantially independent for much longer. I think beali probably deserves at least subspecies status.
- Cape Verde Storm-Petrel, Thalobata jabejabe
- “Pacific Storm-Petrel”, Thalobata cryptoleucura
- Monteiro's Storm-Petrel, Thalobata monteiroi
- Band-rumped Storm-Petrel / Madeiran Storm-Petrel, Thalobata castro
- Least Storm-Petrel, Halocyptena microsoma
- Wedge-rumped Storm-Petrel, Halocyptena tethys
- Black Storm-Petrel, Halocyptena melania
- Matsudaira's Storm-Petrel, Halocyptena matsudairae
- European Storm-Petrel / British Storm-Petrel, Hydrobates pelagicus
- Fork-tailed Storm-Petrel, Hydrobates furcatus
- Ringed Storm-Petrel / Hornby's Storm-Petrel, Cymochorea hornbyi
- Markham's Storm-Petrel, Cymochorea markhami
- Tristram's Storm-Petrel, Cymochorea tristrami
- Guadalupe Storm-Petrel, Cymochorea macrodactyla
- Ashy Storm-Petrel, Cymochorea homochroa
- Swinhoe's Storm-Petrel, Cymochorea monorhis
- Leach's Storm-Petrel, Cymochorea leucorhoa
Procellariidae: Petrels, Shearwaters Leach, 1820
16 genera, 98 species HBW-1
 |
| Click for Procellariidae tree |
|---|
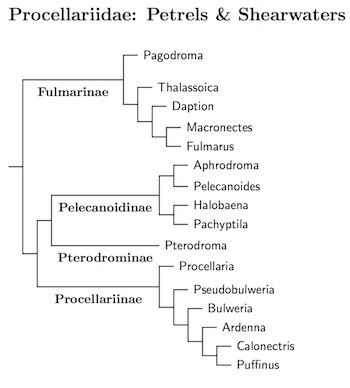
The higher-level relationships within the Procellariidae remain somewhat murky. I've used Prum et al. (2015) as a backbone, and rerooted the tree from Welch et al. (2014) to provide the rest of the structure. The result is also compatible with Gangloff et al. (2012). The taxa shown in brown are subspecies that may deserve species status.
In the new arrangement, the Fulmarinae are the basal group. The Northern Fulmar, Fulmarus glacialis, has been split into the Atlantic Fulmar, Fulmarus glacialis and the Pacific Fulmar, Fulmarus rodgersii, based on Kerr and Dove (2013), who estimated their most recent common ancestor ocurred about 3 million years ago. Although the separation was clear enough in mitochondrial DNA, it didn't show in nuclear DNA. Presumably they simply looked a slow-evolving gene.
Fulmarinae: Fulmars Bonaparte, 1853
- Snow Petrel, Pagodroma nivea
- Antarctic Petrel, Thalassoica antarctica
- Cape Petrel, Daption capense
- Southern Giant-Petrel, Macronectes giganteus
- Northern Giant-Petrel, Macronectes halli
- Southern Fulmar, Fulmarus glacialoides
- Atlantic Fulmar, Fulmarus glacialis
- Pacific Fulmar, Fulmarus rodgersii
Pelecanoidinae: Diving-Petrels and Prions G.R. Gray, 1871 (1850)
The Kerguelen Petrel, Aphrodroma brevirostris is rather uncertainly placed in the basal position here.
The Pelecanoides diving-petrels are traditionally considered a separate family from the petrels (Procellariidae). In many ways, including size, shape, and flight style, they are a southern counterpart of the smaller auks. However, Prum et al. (2015) found them embedded within the Procellariidae.
The Prions and Blue Petrel form the remainder of this group.
- Kerguelen Petrel, Aphrodroma brevirostris
- Peruvian Diving-Petrel, Pelecanoides garnotii
- Common Diving-Petrel, Pelecanoides urinatrix
- South Georgia Diving-Petrel, Pelecanoides georgicus
- Magellanic Diving-Petrel, Pelecanoides magellani
- Blue Petrel, Halobaena caerulea
- Fairy Prion, Pachyptila turtur
- Fulmar Prion, Pachyptila crassirostris
- Broad-billed Prion, Pachyptila vittata
- Salvin's Prion, Pachyptila salvini
- Antarctic Prion, Pachyptila desolata
- Slender-billed Prion, Pachyptila belcheri
Pterodrominae: Gadfly Petrels Verheyen, 1958 (1856)
Among the Petrodroma, I've elevated the Desertas Petrel, Pterodroma desertas to species status based on Zino et al. (2008) and Jesus et al. (2009). Note that the extinct St. Helena Petrel, Pterodroma rupinarum has now been sequenced by Welch et al. (2014) and found to belong to Pterodroma rather than Pseudobulweria.
More recently, the Gray-faced Petrel, Pterodroma gouldi, has been split from Great-winged Petrel, Pterodroma macroptera. See Woods et al. (2016). Note that these are not sister taxa.
- Bonin Petrel, Pterodroma hypoleuca
- Gould's Petrel, Pterodroma leucoptera
- Collared Petrel, Pterodroma brevipes
- Cook's Petrel, Pterodroma cookii
- Masatierra Petrel / De Filippi's Petrel, Pterodroma defilippiana
- Stejneger's Petrel, Pterodroma longirostris
- Pycroft's Petrel, Pterodroma pycrofti
- Soft-plumaged Petrel, Pterodroma mollis
- Gray-faced Petrel, Pterodroma gouldi
- Magenta Petrel, Pterodroma magentae
- Phoenix Petrel, Pterodroma alba
- Atlantic Petrel, Pterodroma incerta
- Great-winged Petrel, Pterodroma macroptera
- White-headed Petrel, Pterodroma lessonii
- Black-capped Petrel, Pterodroma hasitata
- Bermuda Petrel / Cahow, Pterodroma cahow
- Zino's Petrel / Madeira Petrel, Pterodroma madeira
- Desertas Petrel, Pterodroma deserta
- Fea's Petrel / Cape Verde Petrel, Pterodroma feae
- St. Helena Petrel, Pterodroma rupinarum
- Black-winged Petrel, Pterodroma nigripennis
- Chatham Petrel, Pterodroma axillaris
- Providence Petrel, Pterodroma solandri
- Trindade Petrel, Pterodroma arminjoniana
- Kermadec Petrel, Pterodroma neglecta
- Henderson Petrel, Pterodroma atrata
- Herald Petrel, Pterodroma heraldica
- Mottled Petrel, Pterodroma inexpectata
- Murphy's Petrel, Pterodroma ultima
- Barau's Petrel, Pterodroma baraui
- Galapagos Petrel, Pterodroma phaeopygia
- Hawaiian Petrel, Pterodroma sandwichensis
- Juan Fernandez Petrel, Pterodroma externa
- Vanuatu Petrel, Pterodroma occulta
- White-necked Petrel, Pterodroma cervicalis
Procellariinae: Petrels and Shearwaters Leach, 1820
The division of Puffinus into species is based on Austin et al. (2004). Since it is doubtful that the two clades of Puffinus (here called Ardenna and Puffinus) are more closer related to each other than to Calonectris, they are placed in separate genera.
The Calonectris shearwaters have been studied by Gómez-Díaz et al. (2006). They found that the three Atlantic taxa, borealis, diomedea, and edwardsii, form distinct clades that are roughly equidistant genetically, with diomedea perhaps closer to edwardsii. Their study of morphology found diomedea and borealis very close, with edwardsii somewhat more distant. I've treated this as an unresolved trichotomy on the tree. Following the recommendations of Sangster et al. (2012), the three Atlantic taxa are considered distinct species. The Mediterranean population takes the name Scopoli's Shearwater, Calonectris diomedea, the Cape Verde population becomes Cape Verde Shearwater, Calonectris edwardsii, while Cory's Shearwater is now restriced to Calonectris borealis.
The Cory's/Scopoli's split is of potential interest in the ABA area as there are several specimens of Scopoli's from New York in the early 20th century (Bull, 1974). More recently, Scopoli's has been photographed off the North Carolina and Florida coasts.
That brings us to the Puffinus species swamp. Although Austin et al. (2004) went a long way toward clarifying matters, not all of their results were conclusive, and an inability to extract DNA from certain specimens meant that some taxa were not included (specimens of auricularis, bannermani, and gunax did not yield usable DNA, while heinrothi was not sampled at all). They only examined a single gene: cytochrome-b. Although cytochrome-b is usually pretty reliable at this level of analysis, we would be happier if it were confirmed by a multi-gene analysis. Moreover, some clades have weak support, and additional genes might clarify the situation there.
Several extinct Puffinus taxa have been identified. Olson (2010) makes a strong osteological case that fossil bones from Bermuda previously named P. parvus actually belong to Boyd's Shearwater, P. boydi. It appears likely it was extirpated from Bermuda following human occupation. Interestingly, Audubon's Shearwater then briefly colonized the island, but was extirpated in the 20th century. Ramirez et al. (2010) attempted to examine DNA from the extinct Lava Shearwater, P. olsoni, and the Dune Shearwater, P. holeae. Although they were successful with with olsoni, which is probably best regarded as a form of the Manx Shearwater, P. puffinus, they were unsuccessful with holeae.
One interesting thing about the various Puffinus races is the limited overlap in breeding range. Only the Manx Shearwater, P. puffinus even shares an island with other types of Puffinus. This happens even when two or more Puffinus are present in the same area. This helps strengthen the case for species status of a number of races.
Heinroth's Shearwater, Puffinus heinrothi, differs in plumage from most of Puffinus (in our narrow sense). No DNA information is available. It's probably relatively basal and I've listed it first to highlight the uncertainty.
Of the taxa we have DNA for, the Christmas (nativitatis) and Galapagos (subalaris) Shearwaters are basal. They may be more closely related to each other than the rest of Puffinus, but this is not entirely clear (compare Austin et al., 2004 and Ramirez et al., 2010). In any event, the remaining species form a clade, with Hutton's (huttoni) and Fluttering (gavia) Shearwaters of New Zealand grouping together. All of these taxa are monotypic.
The rest of the Puffinus shearwaters are more tightly grouped, but divide into two parts: an Audubon/Manx group and a Tropical Shearwater group. However, there is some ambiguity in the analysis, and the Manx group may actually be basal (or two basal groups). In any event, the inferred timing of the split between Manx (puffinus), Yelkouan (yelkouan), and Balearic (mauretanicus) Shearwaters post-dates the refilling of the Mediterranean Sea about 5 million years ago. Further, the split between the Atlantic Manx/Audubon two clades may have been driven by the closing of the Isthmus of Panama which completed about 3 million years ago. If the isthmus hypothesis is correct, I wonder whether there was one widespread small shearwater prior to the cleavage of the oceans, or whether groups separated on either side of the isthmus then diversified east or west into their respective seas, finally meeting again near the Tropic of Capricorn north of New Zealand.
Besides the Manx group, the Atlantic part includes the Little/Audubon's group. The southerly Little Shearwater (assimilis), includes assimilis, tunneyi, kermadecensis, and haurakiensis. The subspecies elegans has been raised to species level as Subantarctic Shearwater. The northerly Audubon's group includes 4 taxa: Audubon's Shearwater (P. lherminieri lherminieri and P. l. loyemilleri (if valid)), Barolo Shearwater (P. baroli, and Boyd's Shearwater, P. boydi.
The last group contains the rest of the shearwaters. Before proceeding, we consider the Townsend's Shearwater complex, which has been studied by Martínez Gómez et al. (2015). They found that auricularis and newelli are not genetically distinct. Accordingly, Newell's Shearwater, Puffinus newelli, is lumped into Townsend's Shearwater, Puffinus auricularis. However, the third subspecies, myrtae, is sufficiently distinct to elevate to a species, Rapa Shearwater, Puffinus myrtae.
The remaining shearwaters breed in the Indian and Pacific Oceans, from the east coast of Africa to the west coast of the Americas. The first portion is relatively clear-cut. It includes the recently discovered Bryan's Shearwater, Puffinus bryani (Pyle et al., 2011), the monotypic Black-vented (P. opisthomlelas), and Townsend's (P. auricularis, including newelli) Shearwaters, and Rapa Shearwater (P. myrtae).
The 10 remaining taxa appear to be closely related. The unsampled Bannerman's Shearwater, P. bannermani, of Japan is treated as a separate species, as is the Persian Shearwater (P. persicus plus temptator). The remaining races are very closely related and are treated as a single species: Tropical Shearwater, P. bailloni. However, this species is sometimes split further into a Pacific group, Atoll Shearwater (dichrous, plus polynesiae, colstoni, nicolae, and presumably gunax), leaving bailloni and the possibly redundant atrodorsalis as Baillon's Shearwater.
- Gray Petrel, Procellaria cinerea
- White-chinned Petrel, Procellaria aequinoctialis
- Spectacled Petrel, Procellaria conspicillata
- Parkinson's Petrel / Black Petrel, Procellaria parkinsoni
- Westland Petrel, Procellaria westlandica
- Mascarene Petrel, Pseudobulweria aterrima
- Fiji Petrel, Pseudobulweria macgillivrayi
- Tahiti Petrel, Pseudobulweria rostrata
- Beck's Petrel, Pseudobulweria becki
- Bulwer's Petrel, Bulweria bulwerii
- Olson's Petrel, Bulweria bifax
- Jouanin's Petrel, Bulweria fallax
- Pink-footed Shearwater, Ardenna creatopus
- Flesh-footed Shearwater, Ardenna carneipes
- Wedge-tailed Shearwater, Ardenna pacifica
- Buller's Shearwater, Ardenna bulleri
- Short-tailed Shearwater, Ardenna tenuirostris
- Sooty Shearwater, Ardenna grisea
- Great Shearwater, Ardenna gravis
- Streaked Shearwater, Calonectris leucomelas
- Cory's Shearwater, Calonectris borealis
- Scopoli's Shearwater, Calonectris diomedea
- Cape Verde Shearwater, Calonectris edwardsii
- Heinroth's Shearwater, Puffinus heinrothi
- Christmas Shearwater / Kirimati Shearwater, Puffinus nativitatis
- Galapagos Shearwater, Puffinus subalaris
- Fluttering Shearwater, Puffinus gavia
- Hutton's Shearwater, Puffinus huttoni
- Manx Shearwater, Puffinus puffinus
- Balearic Shearwater, Puffinus mauretanicus
- Yelkouan Shearwater, Puffinus yelkouan
- Little Shearwater, Puffinus assimilis
- Subantarctic Shearwater, Puffinus elegans
- Audubon's Shearwater, Puffinus lherminieri
- Barolo Shearwater, Puffinus baroli
- Boyd's Shearwater, Puffinus boydi
- Bryan's Shearwater, Puffinus bryani
- Black-vented Shearwater, Puffinus opisthomelas
- Townsend's Shearwater, Puffinus auricularis
- Rapa Shearwater, Puffinus myrtae
- Persian Shearwater, Puffinus persicus
- Tropical Shearwater, Puffinus bailloni
- Bannerman's Shearwater, Puffinus bannermani