COLIIFORMES Murie, 1872
The Coliiformes are a relict Afrotropical group consisting of 6 species in 2 genera. It wasn't always so. There's a fairly extensive fossil record from Europe and North America. Primitive Coliiformes (Sandcoleidae) are known from the early Eocence (48-56 mya). By the late Eocene, only modern forms are known (Mayr, 2009).
Coliidae: Mousebirds Sundevall, 1836
2 genera, 6 species HBW-6
- Blue-naped Mousebird, Urocolius macrourus
- Red-faced Mousebird, Urocolius indicus
- Speckled Mousebird, Colius striatus
- White-headed Mousebird, Colius leucocephalus
- Red-backed Mousebird, Colius castanotus
- White-backed Mousebird, Colius colius
LEPTOSOMIFORMES Sharpe, 1891
The Cuckoo Roller was originally considered a cuckoo, some affinities with the rollers were noted, and it has more recently been considered its own family, Leptosomidae. Mayr (2008a) discusses some of the differences between the Cuckoo Roller and the true rollers. Darren Naish's comments on the Cuckoo Roller at Tetrapod Zoology are also worth reading.
This is another relict group, consisting of a single species from Madagascar and the Comoros. Fossils are again know from Europe and North America in the early and middle Eocene.
Leptosomidae: Cuckoo Roller Blyth, 1838
1 genus, 1 species HBW-6
- Cuckoo Roller, Leptosomus discolor
TROGONIFORMES AOU, 1886
The name has been attributed to Fürbringer, 1888, but it was already used in 1886 in the 1st AOU checklist.
One unusual feature of trogons is their heterodactyl foot. In some cases this allows fossils to be easily identified as trogoniformes. The trogon lineage seems to date to the late Paleocene (Jarvis et al., 2014; Prum et al., 2015) and early trogoniform fossils have been found from the London Clay and Danish Fur Formations, both from the early Eocene (Mayr, 2009). There may also be an early Eocene fossil from the Green River Formation in North America. When we see a possible Laurasian distribution like this we should keep in mind that there were land bridges between Europe and America in the Eocene (maps may be found in Brikiatis, 2014). We should also keep in mind that the fossil record of Paleogene birds is much better in Europe and North America than in other locations, and that the pattern we see could reflect patterns of preservation and discovery rather than the actual distribution. Nonetheless, our best guess is that trogons originated in Laurasia, likely in Europe.
In spite of their ancient lineage, only one early trogon left any surviving descendants. The crown group of trogons diversified starting in the Miocene, roughly 20 mya (Oliveros, 2015; Prum et al., 2016).
The geographic origin of the crown trogons is unknown, and investigation of their origin has been hampered by a lack of a solidly established phylogeny. Papers by Espinosa de los Monteros (1998, 2000), Johansson and Ericson (2005), Moyle (2005), Ornelas et al. (2009), and Hosner et al. (2010) have put forth a variety of arrangements. Oliveros's Ph.D. dissertation (2015) seems to have resolved the phylogenetic problems. The African trogons (Apaloderma) are the basal branch, the next split is between the Asian trogons and the New World trogons, which are sister clades.
So how do we get a distribution like that, keeping in mind that stem trogons had a European or perhaps Laurasian distribution? We also have the information that the Trogoniformes are sister to the Bucerotiformes-Coraciiformes-Piciformes clade and that the Leptosomiformes are basal to all four orders. This fits in well with the picture of Trogoniformes as having a Laurasian distribution. Oliveros also considers the Coliiformes as part of the picture, but Suh et al. (2015) ans Suh (2016) suggest that the Coliiformes are more distant relatives, leaving the Strigiformes as the next nearest clade.
From a Laurasian distribution, it is easy to imagine some trogons moving to Africa, and then an Asian clade splitting across the Bering Striat (or landbridge) to the Americas, followed by a southward movement perhaps driven by climate change. This is basically the scenario presented by Oliveros (2015).
Trogonidae: Trogons Lesson, 1828
7 genera, 44 species HBW-6
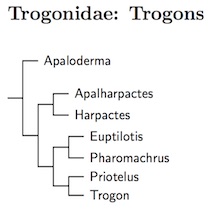 |
| Click for Trogonidae tree |
|---|
The AOU's South American checklist committee had considered making some changes in Trogon species in 2003, partly due to the treatment in Ridgely and Greenfield (2001) and Hilty (2003). For more information, read the discussion for viridis, violaceus, and melanurus on the SACC site. They found that the relevant data, if it existed, was not collected together in a way that allowed them to judge it properly. The publication of DaCosta and Klicka (2008) has changed the balance of evidence here. Although the SACC had not then acted on it, I changed generic limits accordingly.
The species and species groups affected are White-tailed Trogon (T. viridis), Violaceous Trogon (T. violaceus), Black-tailed Trogon (T. malanurus), and Collared/Orange-bellied Trogon (T. collaris and aurantiiventris). The table below summarizes the splits, including genera. The species affected are maked with an asterisk. The question marks on T. macroura (sometimes called Large-tailed Trogon) and T. melanopterus reflect the possibility of future splits.
The SACC has passed (Apr. 8, 2009) a set of proposals concerning splitting mesurus from melanurus, chionurus from viridis, and caligatus from violaceus. I've updated the table with the SACC English names. The NACC has also tentatively ratifed the relevant portions of these changes. The SACC has now recognized the Amazonian Trogon, Trogon ramonianus, as a separate species (Oct 6, 2010). They also adopted the English name Guianan Trogon for the now monotypic T. violacenous. Note that there are still some unresolved issues concerning the exact distribution of these species.
| Species | Subspecies | |
|---|---|---|
| Collared/Orange-bellied Trogon | ||
| *Collared Trogon, T. collaris | extimus, hoethinus, virginalis, subtropicalis, exoptatus, collaris, castaneus | |
| *Orange-bellied Trogon, T. puella | puella, underwoodi, aurantiiventris, flavidor | |
| Black-tailed Trogon Complex | ||
| Lattice-tailed Trogon, T. clathratus | clathratus | |
| *Ecuadorian Trogon, T. mesurus | mesurus | |
| *Black-tailed Trogon, T. melanurus | macroura?, eumorphus, occidentalis, melanurus | |
| Blue-tailed Trogon, T. comptus | comptus | |
| White-tailed Trogon Complex | ||
| Black-headed Trogon, T. melanocephalus | melanocephalus | |
| Citreoline Trogon, T. citreolus | citreolus, sumichrasti | |
| *Green-backed Trogon, T. viridis | viridis, melanopterus? | |
| *White-tailed Trogon, T. chionurus | chionurus | |
| Baird's Trogon, T. bairdii | bairdii | |
| Violaceous Trogon Complex | ||
| *Gartered Trogon, T. caligatus | (sallaei aka braccatus), concinnus, caligatus | |
| *Amazonian Trogon, T. ramonianus | ramonianus, crissalis | |
| Surucua Trogon, T. surrucura | aurantius, surrucura | |
| *Guianan Trogon, T. violaceus | violaceus | |
| Blue-crowned Trogon, T. curucui | peruvianus (bolivianus), curucui, behni | |
All of these forms are apparently field-identifiable both by plumage and voice (see Hilty, 2003; Howell and Webb, 1995; Ridgely and Gwynne, 1989; Ridgely and Greenfield, 2001; Stiles and Skutch, 1989), although some of the differences have not been clearly documented.
These changes combine the Middle American Collared Trogons with Orange-bellied Trogon [I'd previously tried using Jalapa Trogon (used by Sibley and Monroe; 1990, 1993), but have decided to revert to Orange-bellied]. Note that the scientific name does not match the AOU because they don't include puella in the Orange-bellied Trogon. I do, and it has priority. Thus the range of Orange-bellied extends from Mexico to western Panama. The Collared Trogons of South America and Eastern Panama retain the name Collared Trogon. The Black-tailed Trogons west of the Andes in Ecuador and Peru become Ecuadorian Trogon. The White-tailed Trogon can be found from Panama to W Ecuador. The rest of the White-tailed Trogons are grouped as the Green-backed Trogon, even in Trinidad (the population in southeast Brazil may be split at some point). Gartered Trogon is present from Mexico into S. America west of the Andes, and in the north into western Venezuela. The Amazonian Trogon is in the Amazon Basin. Finally, the Guianan Trogon is present in eastern Venezuela, the Guianas, Trinidad, and northeastern Brazil.
- Bar-tailed Trogon, Apaloderma vittatum
- Narina Trogon, Apaloderma narina
- Bare-cheeked Trogon, Apaloderma aequatoriale
- Sumatran Trogon, Apalharpactes mackloti
- Javan Trogon, Apalharpactes reinwardtii
- Orange-breasted Trogon, Harpactes oreskios
- Cinnamon-rumped Trogon, Harpactes orrhophaeus
- Scarlet-rumped Trogon, Harpactes duvaucelii
- Malabar Trogon, Harpactes fasciatus
- Diard's Trogon, Harpactes diardii
- Red-headed Trogon, Harpactes erythrocephalus
- Ward's Trogon, Harpactes wardi
- Red-naped Trogon, Harpactes kasumba
- Philippine Trogon, Harpactes ardens
- Whitehead's Trogon, Harpactes whiteheadi
- Eared Quetzal, Euptilotis neoxenus
- Resplendent Quetzal, Pharomachrus mocinno
- Crested Quetzal, Pharomachrus antisianus
- Golden-headed Quetzal, Pharomachrus auriceps
- White-tipped Quetzal, Pharomachrus fulgidus
- Pavonine Quetzal, Pharomachrus pavoninus
- Cuban Trogon, Priotelus temnurus
- Hispaniolan Trogon, Priotelus roseigaster
- Black-throated Trogon, Trogon rufus
- Elegant Trogon, Trogon elegans
- Mountain Trogon, Trogon mexicanus
- Masked Trogon, Trogon personatus
- Collared Trogon, Trogon collaris
- Orange-bellied Trogon, Trogon puella
- Lattice-tailed Trogon, Trogon clathratus
- Ecuadorian Trogon, Trogon mesurus
- Slaty-tailed Trogon, Trogon massena
- Blue-tailed Trogon / Choco Trogon, Trogon comptus
- Black-tailed Trogon, Trogon melanurus
- Black-headed Trogon, Trogon melanocephalus
- Citreoline Trogon, Trogon citreolus
- Green-backed Trogon, Trogon viridis
- Baird's Trogon, Trogon bairdii
- White-tailed Trogon, Trogon chionurus
- Gartered Trogon, Trogon caligatus
- Amazonian Trogon, Trogon ramonianus
- Surucua Trogon, Trogon surrucura
- Guianan Trogon, Trogon violaceus
- Blue-crowned Trogon, Trogon curucui
BUCEROTIFORMES Fürbringer, 1888
The Bucerotiformes are another ancient order. According to Mayr (2009), the only documented early fossil Bucerotiformes seem more closely related to the hoopoes and woodhoopoes. The Messelirrisoridae are known from the London Clay (UK, early Eocene) and Messel (Germany, middle Eocene). He also mentions there may be a not yet described Bucerotiform from the Green River Formation (Wyoming, early Eocene).
Upupidae: Hoopoes Leach, 1820
1 genus, 4 species HBW-6
- Eurasian Hoopoe, Upupa epops
- African Hoopoe, Upupa africana
- Madagascan Hoopoe, Upupa marginata
- St. Helena Hoopoe, Upupa antaios
Phoeniculidae: Woodhoopoes Bonaparte, 1831
2 genera, 9 species HBW-6
- Black Scimitarbill, Rhinopomastus aterrimus
- Common Scimitarbill, Rhinopomastus cyanomelas
- Abyssinian Scimitarbill, Rhinopomastus minor
- Forest Woodhoopoe, Phoeniculus castaneiceps
- White-headed Woodhoopoe, Phoeniculus bollei
- Green Woodhoopoe, Phoeniculus purpureus
- Black-billed Woodhoopoe, Phoeniculus somaliensis
- Violet Woodhoopoe, Phoeniculus damarensis
- Grant's Woodhoopoe, Phoeniculus granti
Bucorvidae: Ground-Hornbills Bonaparte, 1854
1 genus, 2 species Not HBW Family
- Abyssinian Ground-Hornbill, Bucorvus abyssinicus
- Southern Ground-Hornbill, Bucorvus leadbeateri
Bucerotidae: Hornbills Rafinesque, 1815
16 genera, 59 species HBW-6
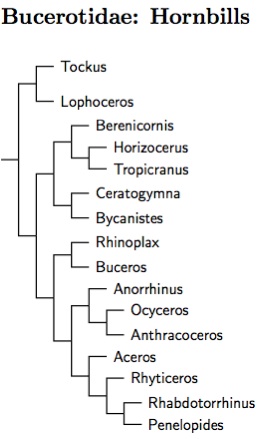 |
| Click for Bucerotidae tree |
|---|
Hornbill phylogeny follows Gonzalez et al. (2013a). They analyzed all hornbill species using both nuclear and mitochondrial DNA. At the genus level, their results match Viseshakul et al. (2011), which used only the cytochrome-b gene. Viseshakul et al. found that much of the hornbill diversity is truly ancient. The genera all seem to have existed as separate lineages for over 30 million years. This caused some problems for them fully resolving the hornbill tree. Gonzalez et al. found a somewhat different arrangement with better support. It is possible that consideration of additional genes would lead to some further adjustment. In particular, I have doubts that Berenicornis (White-crowned Hornbill) is correctly placed yet.
I have followed Hübner et al. (2003) and Gonzalez et al. (2013a) in splitting Tockus into the whistlers (Lophoceros) and cluckers (Tockus). Besides the vocal distinctions, the split bewteen these clades is quite deep. Viseshakul et al. (2011) et al. estimate it at around 45 million years. Note the genus name Lophoceros (Ehrenberg 1833) has priority over Rhynchaceros (Gloger 1842) as suggested by both Hübner et al. (2003) and Gonzalez et al. (2013a). (They subsequently corrected this in Gonzalez et al. 2013b.)
There were two surprises at the species level in Gonzalez et al. (2013). The Black Dwarf-Hornbill, Tockus hartlaubi, does not belong in Tockus. Here it has been placed sister to Tropicranus in the monotypic genus Horizocerus (Oberholser 1899). The Sulawesi Hornbill, Penelopides exarhatus, doesn't belong with Penelopides. Instead, it joins Wrinkled, Walden's, and Writhed Hornbills. Viseshakul et al. (2011) had previously shown they did not belong in Aceros, and I had separated them as Cranobrontes (Riley 1921, type species leucocephalus). But now, since exarhatus has joined them, they must take the name Rhabdotorrhinus (Meyer and Wiglesworth 1898, type exarhatus), which has priority over Cranobrontes.
The genus Aceros has been reduced to a single species. Besides losing 3 species to Rhabdotorrhinus, the other former Aceros hornbill, the Knobbed Hornbill, has been merged into Rhyticeros. It seems to be rather distant from the other Rhyticeros hornbill and could reasonably be put in its own genus, Cranorrhinus (Cabanis and Heine, 1860).
The Red-billed Hornbills are split based on Kemp and Delport (2002) and Delport et al. (2004). Some of the red-billed hornbills were included in Viseshakul et al. (2011), which also supports this treatment.
- Red-billed Dwarf-Hornbill, Lophoceros camurus
- Crowned Hornbill, Lophoceros alboterminatus
- Bradfield's Hornbill, Lophoceros bradfieldi
- African Pied Hornbill, Lophoceros fasciatus
- Hemprich's Hornbill, Lophoceros hemprichii
- African Gray Hornbill, Lophoceros nasutus
- Pale-billed Hornbill, Lophoceros pallidirostris
- Eastern Yellow-billed Hornbill, Tockus flavirostris
- Southern Yellow-billed Hornbill, Tockus leucomelas
- Jackson's Hornbill, Tockus jacksoni
- Von der Decken's Hornbill, Tockus deckeni
- Monteiro's Hornbill, Tockus monteiri
- Damara Red-billed Hornbill, Tockus damarensis
- Southern Red-billed Hornbill, Tockus rufirostris
- Northern Red-billed Hornbill, Tockus erythrorhynchus
- Western Red-billed Hornbill, Tockus kempi
- Tanzanian Red-billed Hornbill, Tockus ruahae
- White-crowned Hornbill, Berenicornis comatus
- Black Dwarf-Hornbill, Horizocerus hartlaubi
- White-crested Hornbill, Tropicranus albocristatus
- Black-casqued Hornbill, Ceratogymna atrata
- Yellow-casqued Hornbill, Ceratogymna elata
- Silvery-cheeked Hornbill, Bycanistes brevis
- Black-and-white-casqued Hornbill, Bycanistes subcylindricus
- Brown-cheeked Hornbill, Bycanistes cylindricus
- White-thighed Hornbill, Bycanistes albotibialis
- Piping Hornbill, Bycanistes fistulator
- Trumpeter Hornbill, Bycanistes bucinator
- Helmeted Hornbill, Rhinoplax vigil
- Rufous Hornbill, Buceros hydrocorax
- Great Hornbill, Buceros bicornis
- Rhinoceros Hornbill, Buceros rhinoceros
- Bushy-crested Hornbill, Anorrhinus galeritus
- Tickell's Brown-Hornbill, Anorrhinus tickelli
- Austen's Brown-Hornbill, Anorrhinus austeni
- Indian Gray-Hornbill, Ocyceros birostris
- Malabar Gray-Hornbill, Ocyceros griseus
- Sri Lanka Gray-Hornbill, Ocyceros gingalensis
- Black Hornbill, Anthracoceros malayanus
- Sulu Hornbill, Anthracoceros montani
- Malabar Pied-Hornbill, Anthracoceros coronatus
- Oriental Pied-Hornbill, Anthracoceros albirostris
- Palawan Hornbill, Anthracoceros marchei
- Rufous-necked Hornbill, Aceros nipalensis
- Knobbed Hornbill, Rhyticeros cassidix
- Sumba Hornbill, Rhyticeros everetti
- Wreathed Hornbill, Rhyticeros undulatus
- Plain-pouched Hornbill, Rhyticeros subruficollis
- Blyth's Hornbill, Rhyticeros plicatus
- Narcondam Hornbill, Rhyticeros narcondami
- Wrinkled Hornbill, Rhabdotorrhinus corrugatus
- Sulawesi Hornbill, Rhabdotorrhinus exarhatus
- Walden's Hornbill, Rhabdotorrhinus waldeni
- Writhed Hornbill, Rhabdotorrhinus leucocephalus
- Visayan Hornbill, Penelopides panini
- Luzon Hornbill, Penelopides manillae
- Mindoro Hornbill, Penelopides mindorensis
- Samar Hornbill, Penelopides samarensis
- Mindanao Hornbill, Penelopides affinis
CORACIIFORMES Forbes, 1884
 |
| Click for Coraciiformes tree |
|---|
The family-level arrangement of the Coraciiformesis based on Ericson et al. (2006a) and Hackett et al. (2008). The Coraciiform families fall into three groups: the bee-eaters (Meropidae); the ground-rollers and rollers (Brachypteraciidae and Coraciidae); the kingfishers, motmots, and todies (Alceidiae, Momotidae, and Todidae). Ericson et al. left the relationships of the three groups unresolved, while Hackett et al. (2008) considered the bee-eaters the basal group, with the roller and kingfisher groups sister. The morphological analysis reported in Mayr (2009) had the bee-eaters grouped with the kingfisher group, and the roller group basal.
Early fossil Coraciiformes include the families Primobucconidae, Eocoraciidae, Geranopteridae, and the species Quasisyndactylus longibranchis. As with many other Afroavian orders, the earliest fossils are from the early Eocene in the Northern Hemisphere (Clarke et al., 2009; Ksepka and Clarke, 2010), in this case including both North America and Europe (Clarke et al., 2009). Although the todies are now restricted to the Caribbean, there are fossils from the Oligocene of Europe as well as North America (Mayr, 2009).
MEROPI Forbes, 1884
Meropidae: Bee-eaters Rafinesque, 1815
3 genera, 27 species HBW-6
The bee-eaters follow Marks et al. (2007).
- Blue-bearded Bee-eater, Nyctyornis athertoni
Click for Meropidae tree - Red-bearded Bee-eater, Nyctyornis amictus
- Purple-bearded Bee-eater, Meropogon forsteni
- Red-throated Bee-eater, Merops bulocki
- White-fronted Bee-eater, Merops bullockoides
- Black-headed Bee-eater, Merops breweri
- Boehm's Bee-eater, Merops boehmi
- Black Bee-eater, Merops gularis
- Blue-moustached Bee-eater, Merops mentalis
- Blue-headed Bee-eater, Merops muelleri
- Little Bee-eater, Merops pusillus
- Blue-breasted Bee-eater, Merops variegatus
- Swallow-tailed Bee-eater, Merops hirundineus
- Cinnamon-chested Bee-eater, Merops oreobates
- Somali Bee-eater, Merops revoilii
- White-throated Bee-eater, Merops albicollis
- Rosy Bee-eater, Merops malimbicus
- Northern Carmine Bee-eater, Merops nubicus
- Southern Carmine Bee-eater, Merops nubicoides
- Green Bee-eater, Merops orientalis
- Chestnut-headed Bee-eater, Merops leschenaulti
- Blue-throated Bee-eater, Merops viridis
- European Bee-eater, Merops apiaster
- Rainbow Bee-eater, Merops ornatus
- Blue-tailed Bee-eater, Merops philippinus
- Blue-cheeked Bee-eater, Merops persicus
- Olive Bee-eater, Merops superciliosus
CORACII Forbes, 1884
Brachypteraciidae: Ground-Rollers Bonaparte, 1854
4 genera, 5 species HBW-6
Kirchman et al. (2001) discuss the split of Geobiastes from Brachypteracias. However, they do not come to a definitive conclusion regarding how the Ground-Rollers are related.
- Short-legged Ground-Roller, Brachypteracias leptosomus
- Scaly Ground-Roller, Geobiastes squamiger
- Long-tailed Ground-Roller, Uratelornis chimaera
- Pitta-like Ground-Roller, Atelornis pittoides
- Rufous-headed Ground-Roller, Atelornis crossleyi
Coraciidae: Rollers Rafinesque, 1815
2 genera, 12 species HBW-6
- Blue-throated Roller, Eurystomus gularis
- Broad-billed Roller, Eurystomus glaucurus
- Dollarbird / Oriental Dollarbird, Eurystomus orientalis
- Purple Roller / Azure Dollarbird, Eurystomus azureus
- Purple Roller, Coracias naevius
- Indian Roller, Coracias benghalensis
- Purple-winged Roller, Coracias temminckii
- Racket-tailed Roller, Coracias spatulatus
- Lilac-breasted Roller, Coracias caudatus
- Abyssinian Roller, Coracias abyssinicus
- European Roller, Coracias garrulus
- Blue-bellied Roller, Coracias cyanogaster
HALCYONI Forbes, 1884
Todidae: Todies Vigors, 1825
1 genus, 5 species HBW-6

The tody sequence is based on Overton and Rhoads (2004). They also considered the todies and motmots sister families, as did Johansson and Ericson (2003). I follow Ericson et al. (2006a) and Hackett et al. (2008), which consider motmots and kingfishers sister families.
- Cuban Tody, Todus multicolor
- Narrow-billed Tody, Todus angustirostris
- Broad-billed Tody, Todus subulatus
- Jamaican Tody, Todus todus
- Puerto Rican Tody, Todus mexicanus
Momotidae: Motmots G.R. Gray, 1840 (1832-33)
6 genera, 15 species HBW-6
The motmot sequence is based on Witt (2004).
The Blue-crowned Motmot complex has been split based on Witt (2004) and Stiles (2009). Witt found that the Andean (Highland) Motmot, Momotus aequatorialis (including chlorolaemus), is not truly part of the Blue-crowned complex, but is sister to the combined Blue-crowned complex plus the Russet-crowned Motmot, Momotus mexicanus.
The Blue-crowned complex proper is split into 6 species based on a combination of Witt (2004) and Stiles (2009). The races are allocated as follows, with approximate species ranges.
| Name | Associated Subspecies | Range |
|---|---|---|
| Blue-capped Motmot, Momotus coeruliceps | coeruliceps | NE Mexico: Nuevo Leon & Tamaulipas |
| Lesson's Motmot, Momotus lessonii | goldmani, exiguus, lessonii | Middle America: Veracruz to W Panama (Chiriqui) |
| Amazonian Motmot, Momotus momota | microstephanus, momota, ignobilis, simplex, cametensis, paraensis, marcgravianus, nattereri, pilcomajensis | Amazon Basin |
| Silver-banded Motmot, Momotus argenticinctus | argenticinctus | W Ecuador and NW Peru, W of Andes |
| Trinidad Motmot, Momotus bahamensis | bahamensis | Trinidad & Tobago |
| Whooping Motmot, Momotus subrufescens | subrufescens, spatha, osgoodi, conexus*, reconditus*, olivaresi* | C & E Panama and NW South America (Chocó, Caribbean slope, Magdalena Valley) |
| * = There is a case for submerging conexus, reconditus, olivaresi into subrufescens. | ||
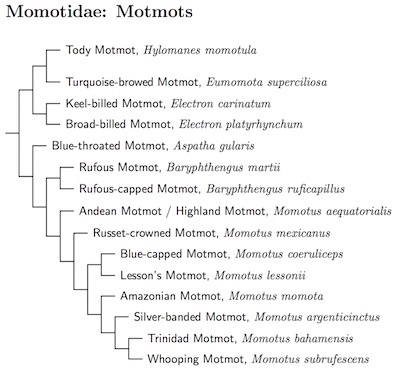
All but the Silver-banded Motmot are recognized by Stiles. However, Witt found it to be sister to the Trinidad/Whooping group, which is why I'm treating it as a separate species.
- Tody Motmot, Hylomanes momotula
- Turquoise-browed Motmot, Eumomota superciliosa
- Keel-billed Motmot, Electron carinatum
- Broad-billed Motmot, Electron platyrhynchum
- Blue-throated Motmot, Aspatha gularis
- Rufous Motmot, Baryphthengus martii
- Rufous-capped Motmot, Baryphthengus ruficapillus
- Andean Motmot / Highland Motmot, Momotus aequatorialis
- Russet-crowned Motmot, Momotus mexicanus
- Blue-capped Motmot, Momotus coeruliceps
- Lesson's Motmot, Momotus lessonii
- Amazonian Motmot, Momotus momota
- Silver-banded Motmot, Momotus argenticinctus
- Trinidad Motmot, Momotus bahamensis
- Whooping Motmot, Momotus subrufescens
Alcedinidae: Kingfishers Rafinesque, 1815
19 genera, 116 species HBW-6
I've followed the taxonomic recommendations of Moyle et al. (2007) for the river kingfishers (Alcedininae). Two subspecies of the Malachite Kingfisher, Corythornis cristata, are sometimes considered full species. They are the Sao Tome Kingfisher (C. cristata thomensis) and the Principe Kingfisher (C. cristata nais). Recent work by Melo and Fuchs (2008) suggests they should continue to be considered subspecies.
The Oriental Dwarf-Kingfisher is here considered to be two species: Black-backed Kingfisher, Ceyx erithaca, and Rufous-backed Kingfisher, Ceyx rufidorsa (including motleyi). See Lim et al. (2010a) for more.
The Silvery Kingfisher, Ceyx argentatus, has been split into Southern Silvery-Kingfisher, Ceyx argentatus, and Northern Silvery-Kingfisher, Ceyx flumenicola. See Collar (2011) and Andersen et al. (2013).
Based on Andersen et al. (2013), the former Variable Dwarf-Kingfisher has been split into 15 allopatric species. Most (all?) of these are field identifiable and the genetic distance between most of them is substantial. They are the species starting at Dimorphic Dwarf-Kingfisher, and continuing to the end of Alcedininae.
Andersen et al. (2015a) examined many of the Todiramphus kingfishers. As a result, the Micronesian Kingfisher, Todiramphus cinnamominus, has been split into Rusty-capped Kingfisher, Todiramphus pelewensis, Guam Kingfisher, Todiramphus cinnamominus (including the extinct miyakoensis), and Pohnpei Kingfisher, Todiramphus reichenbachii.
Although data is lacking, the rearrangment increased the doubt about whether the two taxa united as Tuamotu Kingfisher, Todiramphus gambieri, are really conspecific. They have been split into Mangareva Kingfisher, Todiramphus gambieri, and Niau Kingfisher, Todiramphus gertrudae.
The Collared Kingfisher, Todiramphus chloris, has been split into Collared Kingfisher, Todiramphus chloris, Pacific Kingfisher, Todiramphus sacer, Melanesian Kingfisher, Todiramphus tristrami, Mariana Kingfisher, Todiramphus albicilla, Torresian Kingfisher, Todiramphus sordidus, and Islet Kingfisher, Todiramphus colonus. See the current IOC for the allocation of subspecies.
I've also resequenced Todiramphus based on Andersen et al. (2015a). Seven taxa were not analyzed. Of them, the positioning of Talaud Kingfisher, Todiramphus enigma, Sombre Kingfisher, Todiramphus funebris, and Cinnamon-banded Kingfisher, Todiramphus australasia, is particularly speculative.
Alcedininae: River Kingfishers Rafinesque, 1815
- African Pygmy Kingfisher, Ispidina picta

Click for Alcedininae
and Cerylinae tree - African Dwarf Kingfisher, Ispidina lecontei
- Madagascan Pygmy Kingfisher, Corythornis madagascariensis
- White-bellied Kingfisher, Corythornis leucogaster
- Malachite Kingfisher, Corythornis cristatus
- Malagasy Kingfisher, Corythornis vintsioides
- Blue-banded Kingfisher, Alcedo euryzona
- Blue-eared Kingfisher, Alcedo meninting
- Blyth's Kingfisher, Alcedo hercules
- Common Kingfisher, Alcedo atthis
- Cerulean Kingfisher, Alcedo coerulescens
- Shining-blue Kingfisher, Alcedo quadribrachys
- Half-collared Kingfisher, Alcedo semitorquata
- Sulawesi Dwarf-Kingfisher, Ceyx fallax
- Little Kingfisher, Ceyx pusillus
- Azure Kingfisher, Ceyx azureus
- Bismarck Kingfisher, Ceyx websteri
- Black-backed Kingfisher, Ceyx erithaca
- Rufous-backed Kingfisher, Ceyx rufidorsa
- Philippine Dwarf-Kingfisher, Ceyx melanurus
- Indigo-banded Kingfisher, Ceyx cyanopectus
- Southern Silvery-Kingfisher, Ceyx argentatus
- Northern Silvery-Kingfisher, Ceyx flumenicola
- Dimorphic Dwarf-Kingfisher, Ceyx margarethae
- Sula Dwarf-Kingfisher, Ceyx wallacii
- Buru Dwarf-Kingfisher, Ceyx cajeli
- North Moluccan Dwarf-Kingfisher, Ceyx uropygialis
- Seram Dwarf-Kingfisher, Ceyx lepidus
- New Guinea Dwarf-Kingfisher, Ceyx solitarius
- New Ireland Dwarf-Kingfisher, Ceyx mulcatus
- Manus Dwarf-Kingfisher, Ceyx dispar
- New Georgia Dwarf-Kingfisher, Ceyx collectoris
- Guadalcanal Dwarf-Kingfisher, Ceyx nigromaxilla
- New Britain Dwarf-Kingfisher, Ceyx sacerdotis
- Malaita Dwarf-Kingfisher, Ceyx malaitae
- Bougainville Dwarf-Kingfisher, Ceyx pallidus
- North Solomons Dwarf-Kingfisher, Ceyx meeki
- Makira Dwarf-Kingfisher, Ceyx gentianus
Cerylinae: Water Kingfishers Reichenbach, 1851
- Crested Kingfisher, Megaceryle lugubris
- Giant Kingfisher, Megaceryle maxima
- Belted Kingfisher, Megaceryle alcyon
- Ringed Kingfisher, Megaceryle torquata
- Pied Kingfisher, Ceryle rudis
- Amazon Kingfisher, Chloroceryle amazona
- American Pygmy Kingfisher, Chloroceryle aenea
- Green Kingfisher, Chloroceryle americana
- Green-and-rufous Kingfisher, Chloroceryle inda
Halcyoninae: Tree Kingfishers Vigors, 1825
- Banded Kingfisher, Lacedo pulchella
- Stork-billed Kingfisher, Pelargopsis capensis
- Great-billed Kingfisher, Pelargopsis melanorhyncha
- Brown-winged Kingfisher, Pelargopsis amauroptera
- Glittering Kingfisher, Caridonax fulgidus
- Ruddy Kingfisher, Halcyon coromanda
- White-throated Kingfisher, Halcyon smyrnensis
- Javan Kingfisher, Halcyon cyanoventris
- Chocolate-backed Kingfisher, Halcyon badia
- Gray-headed Kingfisher, Halcyon leucocephala
- Black-capped Kingfisher, Halcyon pileata
- Brown-hooded Kingfisher, Halcyon albiventris
- Striped Kingfisher, Halcyon chelicuti
- Blue-breasted Kingfisher, Halcyon malimbica
- Woodland Kingfisher, Halcyon senegalensis
- Mangrove Kingfisher, Halcyon senegaloides
- Hook-billed Kingfisher, Melidora macrorrhina
- Shovel-billed Kookaburra, Clytoceyx rex
- Spangled Kookaburra, Dacelo tyro
- Rufous-bellied Kookaburra, Dacelo gaudichaud
- Blue-winged Kookaburra, Dacelo leachii
- Laughing Kookaburra, Dacelo novaeguineae
- Lilac Kingfisher, Cittura cyanotis
- Common Paradise-Kingfisher, Tanysiptera galatea
- Kofiau Paradise-Kingfisher, Tanysiptera ellioti
- Biak Paradise-Kingfisher, Tanysiptera riedelii
- Numfor Paradise-Kingfisher, Tanysiptera carolinae
- Little Paradise-Kingfisher, Tanysiptera hydrocharis
- Buff-breasted Paradise-Kingfisher, Tanysiptera sylvia
- Red-breasted Paradise-Kingfisher, Tanysiptera nympha
- Brown-headed Paradise-Kingfisher, Tanysiptera danae
- Green-backed Kingfisher, Actenoides monachus
- Scaly-breasted Kingfisher, Actenoides princeps
- Moustached Kingfisher, Actenoides bougainvillei
- Spotted Wood Kingfisher, Actenoides lindsayi
- Hombron's Kingfisher, Actenoides hombroni
- Rufous-collared Kingfisher, Actenoides concretus
- Yellow-billed Kingfisher, Syma torotoro
- Mountain Kingfisher, Syma megarhyncha
- Blue-black Kingfisher, Todiramphus nigrocyaneus
- Winchell's Kingfisher, Todiramphus winchelli
- Blue-and-white Kingfisher, Todiramphus diops
- Lazuli Kingfisher, Todiramphus lazuli
- Red-backed Kingfisher, Todiramphus pyrrhopygius
- Forest Kingfisher, Todiramphus macleayii
- White-mantled Kingfisher, Todiramphus albonotatus
- Ultramarine Kingfisher, Todiramphus leucopygius
- Chestnut-bellied Kingfisher / Vanuatu Kingfisher, Todiramphus farquhari
- Rusty-capped Kingfisher, Todiramphus pelewensis
- Pacific Kingfisher, Todiramphus sacer
- Marquesan Kingfisher, Todiramphus godeffroyi
- Mewing Kingfisher, Todiramphus ruficollaris
- Society Kingfisher, Todiramphus veneratus
- Chattering Kingfisher, Todiramphus tutus
- Mangareva Kingfisher, Todiramphus gambieri
- Niau Kingfisher, Todiramphus gertrudae
- Guam Kingfisher, Todiramphus cinnamominus
- Pohnpei Kingfisher, Todiramphus reichenbachii
- Flat-billed Kingfisher, Todiramphus recurvirostris
- Collared Kingfisher, Todiramphus chloris
- Talaud Kingfisher, Todiramphus enigma
- Sombre Kingfisher, Todiramphus funebris
- Melanesian Kingfisher, Todiramphus tristrami
- Beach Kingfisher, Todiramphus saurophagus
- Mariana Kingfisher, Todiramphus albicilla
- Cinnamon-banded Kingfisher, Todiramphus australasia
- Torresian Kingfisher, Todiramphus sordidus
- Sacred Kingfisher, Todiramphus sanctus
- Islet Kingfisher, Todiramphus colonus