TELLURAVES Yuri et al., 2013
Telluraves contains slightly more than 3/4 of all bird species. Athough there's some residual uncertainty about the other main branches of Passerea, the land bird clade Telluraves is well-supported by Jarvis et al. (2014) where it receives 100% bootstrap support. Further, Hackett et al. (2008) and Ericson et al. (2006a) both found the hawks and American vultures to be closer to the Picimorphae than to the falcons (see also Suh et al., 2011; McCormack et al., 2013; Yuri et al. 2013). Other papers (e.g., Morgan-Richards et al., 2008) are less supportive, putting all of them somewhere between the Ardeae and the passerines. The order here follows Jarvis et al.
The main division in Telluraves is between the group containing the mousebirds, hawks, owl, and woodpeckers (Afroaves) and the group containing the falcons, parrots, and passerines (Australaves) (Ericson et al., 2006a; Hackett et al., 2008; Ericson, 2012; Jarvis et al., 2014). Australaves is by far the larger group, containing about 64% of all bird species. In contrast, Afroaves only has about 11½% of the species. Still, Afroaves is much larger than any other high-level group than branches off sooner.
I'm not fond of the names Afroaves and Australaves, mainly because the names suggest some knowledge of their origin that we don't have. As you will note in some of comments in the next few pages, some of the groups where the crown group appears to originate in Africa or Australia today (Ericson, 2012) have fossil records that are quite different. The earliest fossils throughout the clade come from the Northern Hemisphere. More precisely, they come from Europe and North America, which were connected in Paleocene and early Eocene.
Afroaves Ericson, 2012
In the TiF list, Afroaves is divided into four groups: Coliiformes, Accipitrimorphae, Strigiformes, and Picimorphae. There is some uncertainty about whether Strigiformes are closer to Accipitrimorphae or to Picimorphae. Suh (2016) and Jarvis et al. (2014) prefer the latter, and that is followed here.
STRIGIFORMES Wagler, 1830
As mentioned earlier, Jarvis et al. (2014) found the Strigiformes closer to the Picimorphae than to the Accipitrimorphae. However, support for this was not that strong, and some trees had the Strigiformes closer to the Accipitrimorphae. For that reason, I'm not including the Strigiformes in either the Accipitrimorphae or Picimorphae at this time.
The fossil record of the owls dates back to the Paleocene, both in Eurpoe and North America, but no Paleogene fossils have been found from the Southern Hemisphere (Mayr, 2009).
Tytonidae: Barn Owls Mathews, 1912 (1860)
2 genera, 19 species HBW-5
Based on Aliabadian et al. (2016), the American Barn-Owl, Tyto furcata (both North and South America, and Hawaii) has been split from Common Barn-Owl / Western Barn-Owl, Tyto alba (Europe, Africa, and the Middle East). Its English name has been simplifed to Western Barn-Owl. The Eastern Barn-Owl, now named Tyto javanica instead of Tyto delicatula, now includes all subspecies from Pakistan and India through Australia and into the Pacific to New Zealand, including stertens.
This split had be hinted at for some time, but there was confusion about which subspecies belonged where. In particular, whether javanica grouped with alba or delicatula. Further spilts may be needed (e.g., Seram Masked Owl, which would be Tyto almae, Jønsson et al, 2016), but this split is all that is entirely clear right now. Some additional species such as Sulawesi Masked-Owl, Tyto rosenbergii are deeply embedded in the Barn-Owl complex.
The current arrangement of the barn owls is based on Uva et al. (2018). Their results suggest that some adjustment to species limits will be needed, but it is not yet clear to me how best to do that.
Following H&M-4 (Dickinson and Remsen, 2013) and König and Weick (2008), the poorly known Congo Bay-Owl has been moved to Tyto from Phodilus. For now, I am treating it as basal within Tyto.
- Oriental Bay-Owl, Phodilus badius
Click for Tytonidae tree - Sri Lanka Bay-Owl, Phodilus assimilis
- Congo Bay-Owl, Tyto prigoginei
- Red Owl, Tyto soumagnei
- Western Barn-Owl, Tyto alba
- American Barn-Owl, Tyto furcata
- Ashy-faced Owl, Tyto glaucops
- Andaman Masked-Owl, Tyto deroepstorffi
- Sulawesi Masked-Owl, Tyto rosenbergii
- Eastern Barn-Owl, Tyto javanica
- Taliabu Masked-Owl, Tyto nigrobrunnea
- Minahassa Masked-Owl, Tyto inexspectata
- African Grass-Owl, Tyto capensis
- Eastern Grass-Owl, Tyto longimembris
- Sooty Owl, Tyto tenebricosa
- Golden Masked-Owl, Tyto aurantia
- Manus Masked-Owl, Tyto manusi
- Moluccan Masked-Owl, Tyto sororcula
- Australian Masked-Owl, Tyto novaehollandiae
Strigidae: Typical Owls Leach, 1820
30 genera, 221 species HBW-5
The overall organization is based on Wink et al. (2008) and Fuchs et al. (2008). The typical owls are divided into three subfamilies: Ninoxinae, Surniinae, and Striginae. These are further divided into tribes, although that is almost overkill as the tribes are barely larger than the main genus, if at all.
Within Ninoxinae, the position of Uroglaux and Sceloglaux is unresolved, although they are believed to be near Ninox on morphological grounds.
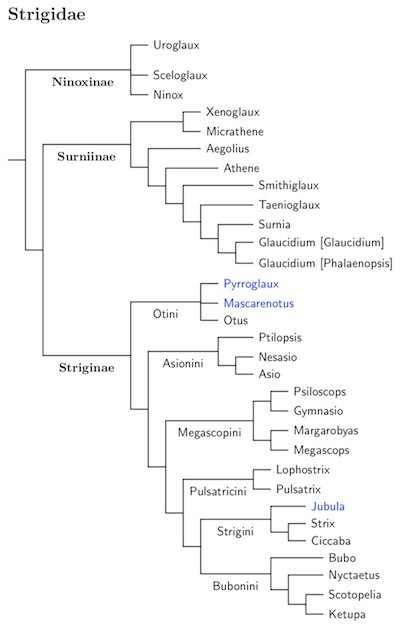 There are four main clades in Surniinae.
There's no molecular evidence concerning Xenoglaux, but it is
believed close to Micrathene, which seems to be basal in Surniinae.
Koparde et al. (2018) found that Heteroglaux is embedded in Athene.
Thus the Forest Owlet becomes Athene blewitti.
There are four main clades in Surniinae.
There's no molecular evidence concerning Xenoglaux, but it is
believed close to Micrathene, which seems to be basal in Surniinae.
Koparde et al. (2018) found that Heteroglaux is embedded in Athene.
Thus the Forest Owlet becomes Athene blewitti.
The treatment of the Glaucidium complex is a bit novel. The basic structure comes from Wink et. al. (2008). Although their taxon sampling is a bit sparse, it suggest five clades in the Glaucidium complex. I've applied the old name Smithiglaux (Bonaparte 1854) to the basal clade, which consists of two African species. The next clade gets the name Taenioglaux, which has recently been revived (e.g., König and Weick, 2008) for Taenioglaux plus Smithiglaux. Next comes the monotypic Surnia, then Glaucidium itself. Glaucidium consists of an Old World clade of owlets (Glaucidium proper) and a New World clade of pygmy-owls. The latter could take the name Phalaenopsis (Bonaparte 1854). For now, I am treating it as a subgenus to maintain consistency with AOU, although a separate genus would make the phylogeny more transparent.
The rest of the owls are in the subfamily Striginae. Otus has been split into the Old World Otini scops-owls and New World Megascopini based on genetic data (e.g., Wink and Heidrich, 1999; Wink et al., 2004b, 2008; Fuchs et al. 2008). Surprisingly, they are not sister groups. Otini consists of the Old World Otus and Pyrroglaux. It is not clear that the latter belongs here, but it seems more likely than the alternatives. The Giant Scops-Owl, Mimizuku gurneyi, is considered part of Otus (Miranda et al., 1997, 2011). I've rearranged Otus some based on Fuchs et al. (2008), Pons et al. (2013), Wink et al. (2008) and HBW-5 (del Hoyo et al., 1999), but a true species-level phylogeny is not available.
The three extinct owls from the Mascarenes are in the genus Mascarenotus, which is thought to be closely related to Otus, or may even be embedded in it.
The Megascopini come after the Asionini. The species arrangement is based on Dantas et al. (2016). They found the Puerto Rican Screech-Owl is sister to Psiloscops, albeit distantly related. Margarobyas is probably close to Megascops. Note that the genus name Gymnoglaux has been replaced by Margarobyas. This is because the type of Gymnoglaux is nudipes, not lawrencii (see Olson and Suárez, 2008). Now that the Puerto Rican Screech-Owl has been moved next to Psiloscops, it takes the genus name Gymnasio (Bonaparte, 1854), which has priority over Gymnoglaux (Cabanis, 1855).
There has been resistance to separating the Flammulated Owl from Otus with Megascops, probably because it is too different from the screech-owls. Nonetheless, the genetic data group it with Megascops, albeit distantly. For that reason it gets its own genus Psiloscops (Coues 1899) as in König and Weick. I place it basally in Megascopini.
Due to a combination of genetic data (Dantas et al., 2016) and vocalizations (König and Weick, 2008), I've split several of the screech-owls. The Vermiculated Screech-Owl, Megascops guatemalae has been split into
- Middle American Screech-Owl, Megascops guatemalae,
- Choco Screech-Owl, Megascops vermiculatus (including centralis),
- Napo Screech-Owl, Megascops napensis, and
- Roraiman Screech-Owl, Megascops roraimae.
The Tawny-bellied Screech-Owl, Megascops watsonii has been split into
- Northern Tawny-bellied Screech-Owl, Megascops watsonii, and
- Southern Tawny-bellied Screech-Owl, Megascops usta.
The recently described Santa Marta Screech-Owl, Megascops gilesi
(Krabbe, 2017) has also been added to the list. All these changes have led to
some rearrangment of the Screech-Owls.
After Pulsatricini, comes Strigini. Jubula is conventionally place here, although its exact affinities remain unclear. My treatment of Strix is a bit unusual. Wink et al. (2008) found two clades in Strix. One is a Holarctic/Old World clade, the other consists of New World species. Some of the New World species have sometimes been separated as Ciccaba, but it has often been objected that they are not distinct from other New World Strix. Here I put all of the New World Strix in an expanded Ciccaba. That leaves the holarctic Great Gray Owl as the only Strix present in the New World. Finally, the African Wood-Owl, Strix woodfordii, which has sometimes been considered close to Ciccaba in the narrow sense, belongs in the Old World clade.
The last clade is Bubonini. DNA is showing that the old generic limits don't work, and the tendency has been to merge everything into Bubo. I think this hides the phylogeny too much. An improvement would be to use two genera, Bubo as constituted here and Ketupa. However, given that some of these species (and even former genera) are mophologically distinct, I think a better solution is to break the clade sister to Bubo into three genera.
We then have 4 genera in Bubonini: Bubo, Nyctaetus (Le Maout 1853), Scotopelia and Ketupa. Ketupa has been expanded with some species that may be closely related (more data is needed). Scotopelia retains its traditional limits. Shelley's and Verreaux's Eagle-Owls are in their own clade, Nyctaetus. It's unclear which clade the Dusky and Akun Eagle-Owls belong in, so I've left them in Bubo for now. Although the Snowy Owl has been considered to be in its own genus, Nyctea, it is actually in Bubo proper, sister to the Great Horned Owl.
The Bermuda Saw-whet Owl, Aegolius gradyi, has been added as it seems to have become extinct in historical times (Olson, 2012).
Northern Boobook, Ninox japonica, and Chocolate Boobook, Ninox randi, have been split from Brown Hawk-Owl, Ninox scutulata, following King (2002).
Rasmussen et al. (2012) re-examined the Philippine Hawk-Owl complex. Their fieldwork turned up two new species in the complex. They argue that the complex consists of 7 species, and I follow their recommendations. This means that Philippine Hawk-Owl is replaced by:
- Luzon Hawk-Owl, Ninox philippensis (including proxima, the doubtful taxon ticaoensis, and centralis
- Mindanao Hawk-Owl, Ninox spilocephala
- Mindoro Hawk-Owl, Ninox mindorensis
- Romblon Hawk-Owl, Ninox spilonotus (including fisheri)
- Cebu Hawk-Owl, Ninox rumseyi
- Camiguin Hawk-Owl, Ninox leventisi
- Sulu Hawk-Owl, Ninox reyi
Guatemalan Pygmy-Owl, Glaucidium cobanense has been split from Northern Pygmy-Owl, Glaucidium gnoma, due to substantial differences in vocalizations (Eisermann and Howell, 2011).
There have been several splits in the scops owls.
- The Negros Scops-Owl, Otus nigrorum, and Everett's Scops-Owl, Otus everetti have been split from Philippine Scops-Owl, Otus megalotis. See Miranda et al. (2011).
- The Arabian Scops-Owl, Otus pamelae, has also been separated from the African Scops-Owl, Otus senegalensis (Pons et al., 2013).
- The Cyprus Scops-Owl, Otus cyprius, has been split from the Eurasian Scops-Owl, Otus scops, based on Flint et al. Zootaxa (2015) and Robb et al. (2015).
- The Socotra Scops-Owl, Otus socotranus, has been separated from the African Scops-Owl, Otus senegalensis, due to substantial differences in vocalizations (e.g., König and Weick, 2008). In fact, its calls are more similar to the Oriental Scops-Owl, Otus sunia. Pons et al. (2013) found it was closely related to O. sunia and O. insularis.
- The newly discovered Rinjani Scops-Owl, Otus jolandae, has been added to the list (Sangster et al., 2013).
Hume's Owl, Strix butleri, has been split into Omani Owl, Strix butleri, and Desert Owl, Strix hadorami based on Robb et al. (2013) and Kirwan et al. (2015). It's clear enough that there are at least two species here. It's somewhat less clear that they are hadorami and butleri, but it seems the best choice given current evidence.
The Cinereous Owl, Ciccaba sartorii has been split from Barred Owl, Ciccaba varia due to difference in both DNA and vocalizations. It is more closely related to the Fulvous Owl than to the Barred Owl (Barrowclough et al., 2011).
Ninoxinae: Hawk-owls Marks et al., 1999
- Papuan Hawk-Owl, Uroglaux dimorpha
- Laughing Owl, Sceloglaux albifacies
- Rufous Owl, Ninox rufa
- Powerful Owl, Ninox strenua
- Little Sumba Hawk-Owl, Ninox sumbaensis
- Morepork, Ninox novaeseelandiae
- Barking Owl, Ninox connivens
- Sumba Boobook, Ninox rudolfi
- Southern Boobook, Ninox boobook
- Northern Boobook, Ninox japonica
- Brown Hawk-Owl, Ninox scutulata
- Chocolate Boobook, Ninox randi
- Togian Boobook, Ninox burhani
- Hume's Hawk-Owl, Ninox obscura
- Andaman Hawk-Owl, Ninox affinis
- Luzon Hawk-Owl, Ninox philippensis
- Mindanao Hawk-Owl, Ninox spilocephala
- Mindoro Hawk-Owl, Ninox mindorensis
- Romblon Hawk-Owl, Ninox spilonotus
- Cebu Hawk-Owl, Ninox rumseyi
- Camiguin Hawk-Owl, Ninox leventisi
- Sulu Hawk-Owl, Ninox reyi
- Ochre-bellied Hawk-Owl / Ochre-bellied Boobook, Ninox ochracea
- Cinnabar Hawk-Owl / Cinnabar Boobook, Ninox ios
- Southern Moluccan Hawk-Owl / Hantu Boobook, Ninox squamipila
- North Moluccan Hawk-Owl / Halmahera Boobook, Ninox hypogramma
- Tanimbar Hawk-Owl / Tanimbar Boobook, Ninox forbesi
- Christmas Island Hawk-Owl / Christmas Boobook, Ninox natalis
- Jungle Hawk-Owl / Papuan Boobook, Ninox theomacha
- Manus Hawk-Owl / Manus Boobook, Ninox meeki
- Speckled Hawk-Owl / Speckled Boobook, Ninox punctulata
- Bismarck Hawk-Owl / New Ireland Boobook, Ninox variegata
- Russet Hawk-Owl / New Britain Boobook, Ninox odiosa
- Solomons Hawk-Owl / Solomons Boobook, Ninox jacquinoti
Surniinae: Owlets and Pygmy-Owls Bonaparte, 1838
- Long-whiskered Owlet, Xenoglaux loweryi
- Elf Owl, Micrathene whitneyi
- Boreal Owl / Tengmalm's Owl, Aegolius funereus
- Northern Saw-whet Owl, Aegolius acadicus
- Bermuda Saw-whet Owl, Aegolius gradyi
- Unspotted Saw-whet Owl, Aegolius ridgwayi
- Buff-fronted Owl, Aegolius harrisii
- Little Owl, Athene noctua
- Spotted Owlet, Athene brama
- Forest Owlet, Athene blewitti
- White-browed Hawk Owl, Athene superciliaris
- Burrowing Owl, Athene cunicularia
- African Barred Owlet, Smithiglaux capensis
- Albertine Owlet, Smithiglaux albertina
- Asian Barred Owlet, Taenioglaux cuculoides
- Javan Owlet, Taenioglaux castanoptera
- Jungle Owlet, Taenioglaux radiata
- Chestnut-backed Owlet, Taenioglaux castanota
- Northern Hawk Owl, Surnia ulula
- Pearl-spotted Owlet, Glaucidium perlatum
- Eurasian Pygmy-Owl, Glaucidium passerinum
- Collared Owlet, Glaucidium brodiei
- Red-chested Owlet, Glaucidium tephronotum
- Sjostedt's Barred Owlet, Glaucidium sjostedti
Subgenus Phalaenopsis
- Northern Pygmy-Owl, Glaucidium gnoma
- Guatemalan Pygmy-Owl, Glaucidium cobanense
- Cuban Pygmy-Owl, Glaucidium siju
- Costa Rican Pygmy-Owl, Glaucidium costaricanum
- Cloud-forest Pygmy-Owl, Glaucidium nubicola
- Andean Pygmy-Owl, Glaucidium jardinii
- Yungas Pygmy-Owl, Glaucidium bolivianum
- Subtropical Pygmy-Owl, Glaucidium parkeri
- Amazonian Pygmy-Owl, Glaucidium hardyi
- Pernambuco Pygmy-Owl, Glaucidium mooreorum
- Least Pygmy-Owl / East Brazilian Pygmy-Owl, Glaucidium minutissimum
- Austral Pygmy-Owl, Glaucidium nana
- Central American Pygmy-Owl, Glaucidium griseiceps
- Tamaulipas Pygmy-Owl, Glaucidium sanchezi
- Colima Pygmy-Owl, Glaucidium palmarum
- Peruvian Pygmy-Owl / Pacific Pygmy-Owl, Glaucidium peruanum
- Ferruginous Pygmy-Owl, Glaucidium brasilianum
Striginae Leach, 1820
Otini: Scops-Owls Bonaparte, 1854
- Palau Owl, Pyrroglaux podargina
- Reunion Owl, Mascarenotus grucheti
- Mauritius Owl, Mascarenotus sauzieri
- Rodrigues Owl, Mascarenotus murivorus
- Sandy Scops-Owl, Otus icterorhynchus
- Sokoke Scops-Owl, Otus ireneae
- White-fronted Scops-Owl, Otus sagittatus
- Reddish Scops-Owl, Otus rufescens
- Serendib Scops-Owl, Otus thilohoffmanni
- Andaman Scops-Owl, Otus balli
- Flores Scops-Owl, Otus alfredi
- Mountain Scops-Owl, Otus spilocephalus
- Rajah Scops-Owl, Otus brookii
- Javan Scops-Owl, Otus angelinae
- Mentawai Scops-Owl, Otus mentawi
- Indian Scops-Owl, Otus bakkamoena
- Collared Scops-Owl, Otus lettia
- Sunda Scops-Owl, Otus lempiji
- Palawan Scops-Owl, Otus fuliginosus
- Japanese Scops-Owl, Otus semitorques
- Giant Scops-Owl, Otus gurneyi
- Negros Scops-Owl, Otus nigrorum
- Philippine Scops-Owl, Otus megalotis
- Everett's Scops-Owl, Otus everetti
- Arabian Scops-Owl, Otus pamelae
- Wallace's Scops-Owl, Otus silvicola
- Eurasian Scops-Owl, Otus scops
- Cyprus Scops-Owl, Otus cyprius
- Sao Tome Scops-Owl, Otus hartlaubi
- Pemba Scops-Owl, Otus pembaensis
- African Scops-Owl, Otus senegalensis
- Pallid Scops-Owl, Otus brucei
- Mindanao Scops-Owl, Otus mirus
- Luzon Scops-Owl, Otus longicornis
- Mindoro Scops-Owl, Otus mindorensis
- Rinjani Scops-Owl, Otus jolandae
- Moluccan Scops-Owl, Otus magicus
- Sula Scops-Owl, Otus sulaensis
- Siau Scops-Owl, Otus siaoensis
- Mantanani Scops-Owl, Otus mantananensis
- Ryukyu Scops-Owl, Otus elegans
- Sulawesi Scops-Owl, Otus manadensis
- Sangihe Scops-Owl, Otus collari
- Biak Scops-Owl, Otus beccarii
- Simeulue Scops-Owl, Otus umbra
- Enggano Scops-Owl, Otus enganensis
- Nicobar Scops-Owl, Otus alius
- Torotoroka Scops-Owl, Otus madagascariensis
- Rainforest Scops-Owl, Otus rutilus
- Mayotte Scops-Owl, Otus mayottensis
- Karthala Scops-Owl, Otus pauliani
- Moheli Scops-Owl, Otus moheliensis
- Anjouan Scops-Owl, Otus capnodes
- Seychelles Scops-Owl, Otus insularis
- Socotra Scops-Owl, Otus socotranus
- Oriental Scops-Owl, Otus sunia
Asionini: Eared Owls Vigors, 1825
- Northern White-faced Owl, Ptilopsis leucotis
- Southern White-faced Owl, Ptilopsis granti
- Fearful Owl, Nesasio solomonensis
- Short-eared Owl, Asio flammeus
- Marsh Owl, Asio capensis
- Striped Owl, Asio clamator
- Stygian Owl, Asio stygius
- Jamaican Owl, Asio grammicus
- Long-eared Owl, Asio otus
- Abyssinian Owl, Asio abyssinicus
- Madagascan Owl, Asio madagascariensis
Megascopini: Screech-Owls Wink et al., 2008? Informal?
Both Megascopini and Pulsatrigini have been used by Wink et al. (2008), but do not seem to have been designated as new taxa, which is now required by the ICZN (since 1999). The spelling of Pulsatrigini has been corrected to Pulsatricini.
- Flammulated Owl, Psiloscops flammeolus
- Puerto Rican Screech-Owl, Gymnasio nudipes
- Bare-legged Owl, Margarobyas lawrencii
- Whiskered Screech-Owl, Megascops trichopsis
- Bare-shanked Screech-Owl, Megascops clarkii
- White-throated Screech-Owl, Megascops albogularis
- Tropical Screech-Owl, Megascops choliba
- Koepcke's Screech-Owl, Megascops koepckeae
- Long-tufted Screech-Owl, Megascops sanctaecatarinae
- Bearded Screech-Owl, Megascops barbarus
- Pacific Screech-Owl, Megascops cooperi
- Western Screech-Owl, Megascops kennicottii
- Eastern Screech-Owl, Megascops asio
- Balsas Screech-Owl, Megascops seductus
- Rufescent Screech-Owl, Megascops ingens
- Colombian Screech-Owl, Megascops colombianus
- Cinnamon Screech-Owl, Megascops petersoni
- Cloud-forest Screech-Owl, Megascops marshalli
- Montane Forest Screech-Owl / Yungas Screech-Owl, Megascops hoyi
- Middle American Screech-Owl, Megascops guatemalae
- Choco Screech-Owl, Megascops vermiculatus
- Napo Screech-Owl, Megascops napensis
- Roraiman Screech-Owl, Megascops roraimae
- Santa Marta Screech-Owl, Megascops gilesi
- Peruvian Screech-Owl / West Peruvian Screech-Owl, Megascops roboratus
- Northern Tawny-bellied Screech-Owl, Megascops watsonii
- Southern Tawny-bellied Screech-Owl, Megascops usta
- Black-capped Screech-Owl, Megascops atricapilla
Pulsatricini Wink et al., 2008? Informal?
- Crested Owl, Lophostrix cristata
- Spectacled Owl, Pulsatrix perspicillata
- Tawny-browed Owl, Pulsatrix koeniswaldiana
- Band-bellied Owl, Pulsatrix melanota
Strigini: Wood-Owls Leach, 1820
- Maned Owl, Jubula lettii
- Great Gray Owl, Strix nebulosa
- Ural Owl, Strix uralensis
- Pere David's Owl, Strix davidi
- Tawny Owl, Strix aluco
- Himalayan Owl, Strix nivicolum
- Mottled Wood-Owl, Strix ocellata
- Spotted Wood-Owl, Strix seloputo
- Brown Wood-Owl, Strix leptogrammica
- Omani Owl, Strix butleri
- African Wood-Owl, Strix woodfordii
- Desert Owl, Strix hadorami
- Spotted Owl, Ciccaba occidentalis
- Barred Owl, Ciccaba varia
- Cinereous Owl, Ciccaba sartorii
- Fulvous Owl, Ciccaba fulvescens
- Rusty-barred Owl, Ciccaba hylophila
- Rufous-legged Owl, Ciccaba rufipes
- Chaco Owl, Ciccaba chacoensis
- Mottled Owl, Ciccaba virgata
- Black-and-white Owl, Ciccaba nigrolineata
- Black-banded Owl, Ciccaba huhula
- Rufous-banded Owl, Ciccaba albitarsis
Bubonini: Eagle-Owls Vigors, 1825
- Snowy Owl, Bubo scandiacus
- Great Horned Owl, Bubo virginianus
- Indian Eagle-Owl, Bubo bengalensis
- Spotted Eagle-Owl, Bubo africanus
- Grayish Eagle-Owl, Bubo cinerascens
- Cape Eagle-Owl, Bubo capensis
- Pharaoh Eagle-Owl, Bubo ascalaphus
- Eurasian Eagle-Owl, Bubo bubo
- Dusky Eagle-Owl, Bubo coromandus
- Akun Eagle-Owl, Bubo leucostictus
- Shelley's Eagle-Owl, Nyctaetus shelleyi
- Verreaux's Eagle-Owl, Nyctaetus lacteus
- Pel's Fishing-Owl, Scotopelia peli
- Rufous Fishing-Owl, Scotopelia ussheri
- Vermiculated Fishing-Owl, Scotopelia bouvieri
- Spot-bellied Eagle-Owl, Ketupa nipalensis
- Barred Eagle-Owl, Ketupa sumatrana
- Nduk Eagle-Owl / Usambara Eagle-Owl, Ketupa vosseleri
- Fraser's Eagle-Owl, Ketupa poensis
- Philippine Eagle-Owl, Ketupa philippensis
- Blakiston's Fish-Owl, Ketupa blakistoni
- Brown Fish-Owl, Ketupa zeylonensis
- Tawny Fish-Owl, Ketupa flavipes
- Buffy Fish-Owl, Ketupa ketupu
PICIMORPHAE
Other than their ordering and associationg with the Piciformes, the treatment of much of the remaining non-passerine families is close to that of the Sibley-Monroe list. Changes over time have mostly involved whether to consider certain groups families or subfamilies.
The mousebirds, cuckoo roller, and trogons are placed in separate orders, as their affinities with the other Picimorphae are relatively distant. The hornbills, which are split into Bucorvidae (ground-hornbills) and Bucerotidae (hornbills) form a grouping with the hoopoes and woodhoopoes. The bee-eaters, rollers, ground-rollers, todies, motmots, and kingfishers form the Coraciiformes. The last group is the the Piciformes, which I take to include the “Galbuliformes”. They will be considered on a later page.
Jarvis et al. (2014) estimated the split between the Coraciiformes and Piciformes at around 40 mya. If their estimates are correct, this is by far the most recent ordinal-level split. If the estimate holds up, it might be worth thinking about merging them into one order. Indeed, even if the hornbills are also included, it would still be one of the most recent ordinal-level splits. There is some fossil evidence (Primobucco) that suggests the split occurred earlier (e.g., Clarke et al., 2009; Ksepka and Clarke, 2010). Or perhaps Primobucco, which was originally considered a puffbird, is from before the split between the two groups. This has to be considered due to differences between phylogenetic analysis of Clarke et al. (2009) and the DNA evidence.