Passerid Clade
While the Estrilid clade is primarily old world and southern, its Passerid sister clade is cosmopolitan. More precisely, the nine-primaried oscinces are cosmopolitan, while their sister group, the Passeridae, are restricted to the Old World.
Passeridae: Old World Sparrows, Snowfinches Rafinesque, 1815
8 genera, 43 species HBW-15
The Passeridae are seed-eating birds, primarily distributed in the southern portion of the Palearctic, but ranging into the Afrotropics. Several passerids are closely associated with humans, especially the ubiquitous House Sparrow, which has spread worldwide.
Dickinson et al. (2003) considered Plocepasser, Histurgops, Pseudonigrita, and Philetairus to be Passeridae. However, Groth (1998) found them to be in the Ploceidae.
Cinnamon Ibon: The most astonishing change in the Passeridae is due to Fjeldså et al. (2010). They found that the Cinnamon Ibon, long thought to be an abberant white-eye, is actually a sparrow. This is a canopy bird from the cloud-forest of Mindanao, in the Philippines. It's a long way from anything we think of as a sparrow. It's also a long way physically. The nearest native populations of any sparrows are on the Asian mainland. Nonetheless, it is a sparrow, or at least a close relative. Indeed, Fjeldså et al. (2010) report than its skull is similar to a sparrow, and that it has many other features in common with the other Passeridae besides DNA. Even so, the Ibon is only a distant relative of the other Passeridae. Their most recent common ancestors lived about 17.5 mya (Päckert et al., 2021), only a short time before the split of the Passeridae and Motacillidae. As a result I'm treating the Ibon is a subfamily of Passeridae.
Pale Rockfinch: Interestingly enough, the Pale Rockfinch (Carpospiza) appears to be only distantly related to the other Passeridae. This was hinted at some time ago, and it had been suggested by H. Mendelssohn (fide HBW-14) that it did not even belong in Passeridae. The question of its affinities were also addressed by Bock (2004), Bock and Morony (1978a, b), and by Ivanitskii (1997). Bock and Morony identified features of the tongue that separate Passeridae from closely related families. These special features are connected with the ability to easily husk seeds. Ivanitskii (1997) and later Bock (2004) were able to confirm that Carpospiza shares this feature.
There are samples of the 16S and cox-1 genes from Carpospiza. They were used in an unpublished tree by Raty. It appears to be one of the basal taxa in Passeridae, along with the Cinnamon Ibon (see below). However, its position was not fully resolved and support was very weak.
The Pale Rockfinch may be as distant from the other Passeridae as the Ibon is, with common ancestors about 17.5 mya (Päckert et al., 2021). This is consistent with the date of about 19 mya for the Passeridae-Motacillidae split found by Oliveros et al. (2019). The result of all this is that I'm treating both as subfamilies of Passeridae until more information is available. I have however, moved Carpospiza slightly, so that it is now basal within Passeridae, rather than following
Fjeldså et al. (2020), Figure 14-38, present a phylogeny where Carpospiza is the basal member Motacillidae (wagtails and pipits). However, it is not included in their Appendix 2. They don't given detailed information about why they think Carpospiza belongs in Motacillidae. The only hint Fjeldså et al. give is to cite Oliveros et al. (2019) in support of the idea that Carpospiza and Motacillidae (wagtails and pipits) have a common ancestor. But that is true of all of the Passeridae! Oliveros et al. do not explicitly mention Carpospiza. Where Fjeldså et al. got the idea that Carpospiza belongs in Motacillidae is a mystery to me. Given the differences between the Pale Rock Sparrow and the wagtails and pipits, including the only available genetic evidence, I think Fjeldså et al. are mistaken.
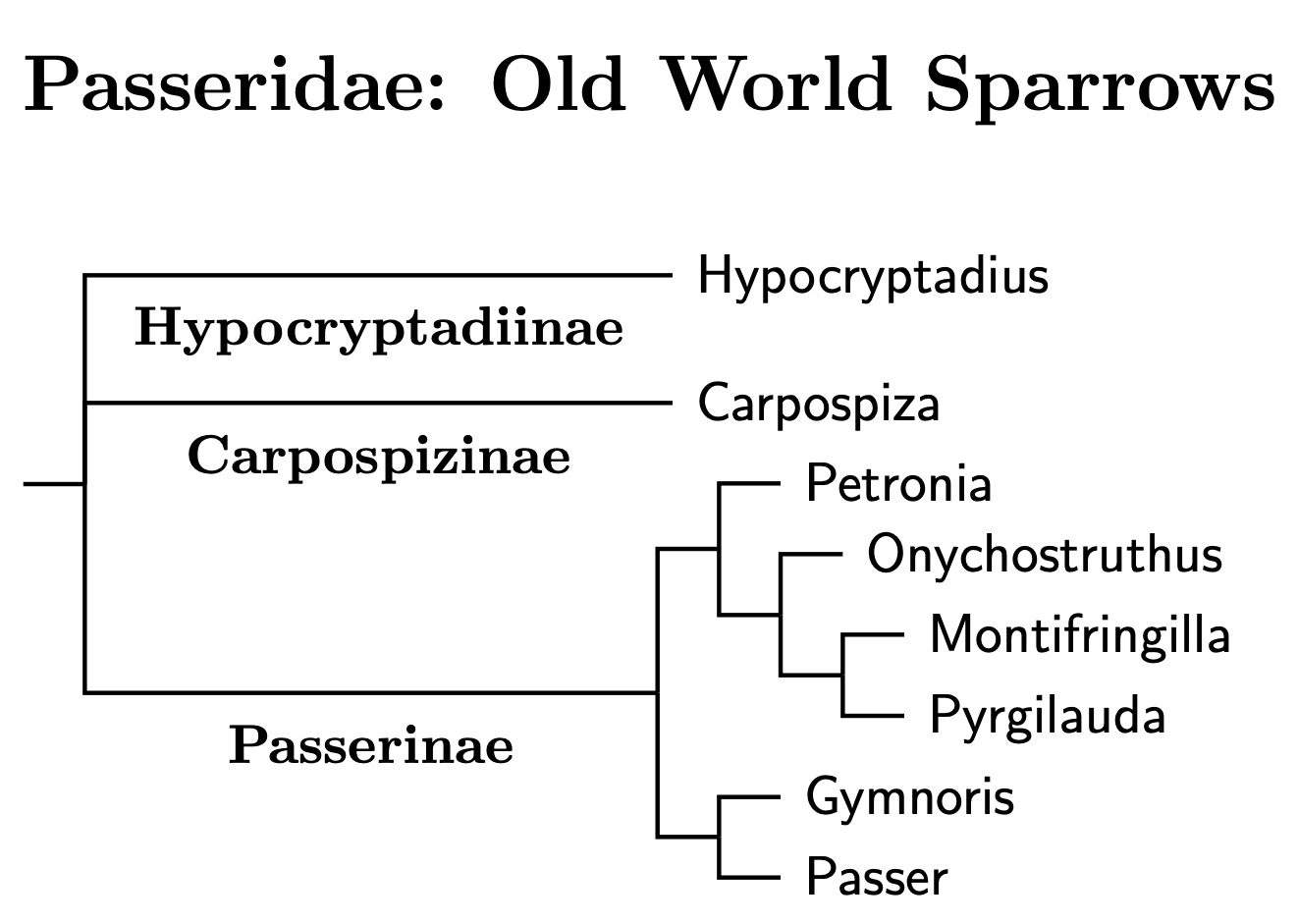 |
| Click for Passeridae species tree |
|---|
The current phylogeny for Passeridae is based on Päckert et al. (2021). Early versions of TiF relied on Price et al. (2014), which included one species of Gymnoris in their analysis as well as Qu et al. (2006), who examined the snowfinches. The genus tree, which remains the same, was initially based on a combination of Fjeldså et al. (2010), Price et al. (2014), Qu et al. (2006) and Raty. Earlier notions of relations within Passer itself have now been substantially altered by Päckert et al.'s (2021) study of Passeridae. They sequenced 18/28 species of genus Passer and 75% of the entire Passeridae family.
Passeridae — Additional Taxonomic Notes
Tibetan Snowfinches: Unfortunately, the name Tibetan Snowfinch has been moved from Montifringilla adamsi to Montifringilla henrici. To avoid confusion, I'm keeping Henri's Snowfinch as the primary name for M. henrici.
Bush-Sparrows: The secondary English names of Gymnoris involving Petronia have been removed as these birds have been separated from the genus Petronia.
Russet Sparrow: The specific epithet for Russet Sparrow has been changed from rutilans Temminck 1836 to cinnamomeus Gould 1836 (not 1838) due to priority.
Zarudny's Sparrow: The Asian subspecies Zarudny's Sparrow, Passer zarudnyi, has been split from Desert Sparrow, Passer simplex, which now contains only the African subspecies (Kirwan et al., 2009).
Italian Sparrow: The Italian Sparrow, Passer italiae, is recognized as a separate species based on Hermansen et al. (2011) and Elvgin et al. (2011). Although Töpfer (2006) had argued that it was a subspecies of the Spanish Sparrow, based on apparent ongoing hybridization, Hermansen et al. (2011) find that this is not the case. Rather, their genetic data found that the sympatric populations in Italy's Gargano Peninsula showed no evidence of gene flow. However, they did find a hybrid zone with the House Sparrow in the Alps. The hybrid zone appears narrow and relatively stable, and does not suggest substantial ongoing gene flow (and they did not find evidence of any). The twin papers produced by a group at Oslo, Hermansen et al. (2011) and Elgvin et al. (2011), explore the evidence that the Italian Sparrow is the result of past hybridization between the House and Spanish Sparrows.
Passeridae: Old World Sparrows, Snowfinches
Hypocryptadiinae: Cinnamon Ibon Hachisuka 1930
- Cinnamon Ibon, Hypocryptadius cinnamomeus
Carpospizinae: Pale Rockfinch Informal
- Pale Rockfinch, Carpospiza brachydactyla
Passerinae: Old World Sparrows, Snowfinches Rafinesque, 1815
- Rock Sparrow, Petronia petronia
- White-rumped Snowfinch, Onychostruthus taczanowskii
- Henri's Snowfinch / Tibetan Snowfinch, Montifringilla henrici
- White-winged Snowfinch, Montifringilla nivalis
- Black-winged Snowfinch, Montifringilla adamsi
- Rufous-necked Snowfinch, Pyrgilauda ruficollis
- Pere David's Snowfinch, Pyrgilauda davidiana
- Afghan Snowfinch, Pyrgilauda theresae
- Blanford's Snowfinch, Pyrgilauda blanfordi
- Yellow-throated Bush-Sparrow, Gymnoris superciliaris
- Sahel Bush-Sparrow, Gymnoris dentata
- Chestnut-shouldered Bush-Sparrow / Yellow-throated Sparrow, Gymnoris xanthocollis
- Yellow-spotted Bush-Sparrow, Gymnoris pyrgita
- Northern Gray-headed Sparrow, Passer griseus
- Swainson's Sparrow, Passer swainsonii
- Swahili Sparrow, Passer suahelicus
- Parrot-billed Sparrow, Passer gongonensis
- Southern Gray-headed Sparrow, Passer diffusus
- Cape Sparrow, Passer melanurus
- Chestnut Sparrow, Passer eminibey
- Kordofan Sparrow, Passer cordofanicus
- Shelley's Sparrow, Passer shelleyi
- Kenya Sparrow, Passer rufocinctus
- Great Sparrow, Passer motitensis
- Russet Sparrow, Passer rutilans
- Eurasian Tree Sparrow, Passer montanus
- Saxaul Sparrow, Passer ammodendri
- Iago Sparrow, Passer iagoensis
- Desert Sparrow, Passer simplex
- Zarudny's Sparrow, Passer zarudnyi
- Dead Sea Sparrow, Passer moabiticus
- Sudan Golden-Sparrow, Passer luteus
- Arabian Golden-Sparrow, Passer euchlorus
- Plain-backed Sparrow, Passer flaveolus
- Abd al Kuri Sparrow, Passer hemileucus
- Socotra Sparrow, Passer insularis
- Sind Sparrow, Passer pyrrhonotus
- House Sparrow, Passer domesticus
- Italian Sparrow, Passer italiae
- Spanish Sparrow, Passer hispaniolensis
- Somali Sparrow, Passer castanopterus
Nine-primaried Oscines
The nine-primaried oscines are called that because they appear to have nine primary feathers. Actually, they have ten primaries, but the tenth primary is reduced and usually hidden under the ninth primary covert (Hall, 2004). The concept was introduced by Wallace (1874), who referred to them as the Tanagroid Passeres. Wallace's grouping was not a natural group. It also included the waxwings, flowerpeckers (in which Wallace included pardalotes and white-eyes), and swallows.
The nine-primaried oscines repesent a recent development, with most of its families originating less than 15 million years ago. Nonetheless, the nine-primaried oscines are extremely numerous, and account for almost 18% of all passeriformes. To organize this better, we will use family-level and genus-level groups that are younger than those in most other parts of the avian tree. However, things will stay pretty much unchanged for species.
The basal division in the nine-primaried oscines is between the Motacillidae and the “finches”, Sibley and Monroe's broadly-defined Fringillidae. Wagtails and pipits are cosmopolitan birds. They are typically open country insectivores.
Motacillidae: Wagtails, Longclaws & Pipits Horsfield, 1821
8 genera, 67 species HBW-9
 |
| Click for Motacillidae tree |
|---|
The overall organization of the Motacillidae is based on a combination of Alström et al. (2015a), Drovetski et al. (2018), Harris et al. (2018), Outlaw and Voelker (2006b), Van Ells and Norambuena (2018), and Voelker and Edwards (1998). When there are disagreements, the more recent papers have been given heavier weight.
One interesting thing in Alström et al. (2015a) is that their multigene analysis yields a different placement of the longclaws compared with Outlaw and Voelker (2006b). The limited taxon sampling leaves some residual uncertainty, but it has caused me to separate some of Anthus in Corydalla (Vigors, 1825, type richardi) and Cinaedium (Sundevall, 1850, type lineiventre).
Sao Tome Shorttail: The Motacillidae also include two species that were long thought to belong to Sylviidae, the Sao Tome Shorttail (Amaurocichla), and the Madanga (Madanga).
The Sao Tome Shorttail (or Bocage's Longbill) was thought to be related to the Macrosphenus longbills. When the old Sylviidae were broken up, that put it in the crombec family, Macrosphenidae. However, Johansson et al. (2008b) results suggested by it was related to the wagtails and pipits, and possibly sister to the wagtails. Alström et al. (2015a) found it was not only related to the wagtails, but is actually part of Motacilla.
Madanga: More surprisingly, Alström et al. (2015a) also found that the Madanga is actually a pipit, not a white-eye (Sylviioidea: Zosteropidae). In fact, it falls in the genus Anthus. Its closest relative seems to be the Alpine Pipit, Anthus gutturalis. Laurent Raty has pointed out a complication. In 1831, Lesson used the name Anthus ruficollis. The Madanga wasn't named until 1923 by Rothschild and Hartert, so Lesson's ruficollis has priority if both species are placed in Anthus. The fact that Lesson's ruficollis is a junior synonym of Anthus cervinus (orginally Motacilla cervina Pallas 1811) does not eliminate the conflict. Until either a new name is proposed or Lesson's name Anthus ruficollis is suppressed, the Madanga won't have a proper scientic name. For the present, I will refer to it as Anthus “ruficollis”.
Wagtails: The wagtails, and particularly the yellow-colored wagtails, are more troublesome. Mitochondrial DNA analysis (Alström and Mild, 2003; Ödeen and Bjöklund, 2003; Outlaw and Voelker, 2006b, Pavlova et al., 2003) suggests that there are two species of Citrine Wagtail (citreola and werae) and three species of Yellow Wagtail (flava, taivana, and tschutschensis). The AOU has adopted the split of the Eastern Yellow Wagtail (tschutschensis), but takes no official position on the 3-way split. Christidis and Boles (2008) accept the 3-way split with Green-headed Yellow Wagtail, Motacilla taivana, being the third species.
These splits are not followed by Alström and Mild (2003) or by Ödeen and Björklund (2003), both of which also consider nuclear DNA. The nuclear DNA yields a different tree for the wagtails, where macronyx, taivana and tschutschensis form a clade that can be regarded as a species: Eastern Yellow Wagtail, Motacilla tschutschensis. This species is sister to the Gray Wagtail, Motacilla cinerea. The citrine wagtails end up as sisters, and can also be regarded as a single species. Ödeen and Björklund (2003) argue that the mitochondrial tree reflects the effects of past hybridization.
In the end, I have gone with the analysis by Harris et al. (2018). This treats the Citrine and Eastern Yellow Wagtails as sister species, with the Western Yellow Wagtail sister to both, and the whole lot sister to the Gray Wagtail. Drovetski et al. (2018) has a bit different arrangment.
Large Pipits: There have also been questions concerning whether the White Wagtail is a single species. Some authorities have separated lugens as Black-backed Wagtail and yarrelli as Pied Wagtail. However, it is hard to make a genetic case for either of these (see Alström and Mild, 2003; Pavlova et al., 2005; Voelker, 2002), with yarrelli ending up near alba and alba and lugens intertwinned.
Australasian Pipit, Corydalla novaeseelandiae, has been split into Australian Pipit, Corydalla australis, and New Zealand Pipit, Corydalla novaeseelandiae. This split was recommended by Schodde and Mason (1999), but rejected by Christidis and Boles (2008) “in the absence of molecular evidence”. Tavares and Baker (2008) provided limited molecular evidence in the form of a barcode divergence of 4.1%, which is a good indication that they are separate species.
Based on Davies and Peacock (2014), the Kimberley Pipit, Corydalla pseudosimilis, is considered conspecific with African Pipit, Corydalla cinnamomea, and the Long-tailed Pipit, Corydalla longicaudatus, is considered conspecific with Buffy Pipit, Corydalla vaalensis. In both cases, the original collections of these species seem to involve several taxa.
New World Pipits: The New World pipits have been rearranged based on Van Ells and Norambuena (2018). Further, their analysis resulted in two splits and one lump.
- Peruvian Pipit, Anthus peruvianus, has been split from Yellowish Pipit, Anthus lutescens
- Puna Pipit, Anthus brevirostris, has been split from Short-billed Pipit, Anthus furcatus.
- South Georgia Pipit, Anthus antarcticus, has been lumped into Correndera Pipit, Anthus correndera due to very small DNA differences between them.
Motacillinae: Wagtails, Longclaws & Pipits Horsfield, 1821
- Forest Wagtail, Dendronanthus indicus
- Mountain Wagtail, Motacilla clara
- Cape Wagtail, Motacilla capensis
- Sao Tome Shorttail, Motacilla bocagii
- Madagascan Wagtail, Motacilla flaviventris
- Gray Wagtail, Motacilla cinerea
- Western Yellow Wagtail, Motacilla flava
- Eastern Yellow Wagtail, Motacilla tschutschensis
- Citrine Wagtail, Motacilla citreola
- White-browed Wagtail, Motacilla maderaspatensis
- Mekong Wagtail, Motacilla samveasnae
- Japanese Wagtail, Motacilla grandis
- White Wagtail, Motacilla alba
- African Pied Wagtail, Motacilla aguimp
- Upland Pipit, Corydalla sylvana
- Australian Pipit, Corydalla australis
- New Zealand Pipit, Corydalla novaeseelandiae
- Tawny Pipit, Corydalla campestris
- Berthelot's Pipit, Corydalla berthelotii
- Richard's Pipit, Corydalla richardi
- Paddyfield Pipit, Corydalla rufula
- Blyth's Pipit, Corydalla godlewskii
- Plain-backed Pipit, Corydalla leucophrys
- Wood Pipit, Corydalla nyassae
- Long-billed Pipit, Corydalla similis
- African Pipit, Corydalla cinnamomea
- Malindi Pipit, Corydalla melindae
- Buffy Pipit, Corydalla vaalensis
- Long-legged Pipit, Corydalla pallidiventris
- Sokoke Pipit, Cinaedium sokokense
- Short-tailed Pipit, Cinaedium brachyurum
- Bushveld Pipit, Cinaedium caffrum
- Mountain Pipit, Cinaedium hoeschi
- Striped Pipit, Cinaedium lineiventre
- African Rock Pipit, Cinaedium crenatum
- Golden Pipit, Tmetothylacus tenellus
- Yellow-breasted Pipit, Hemimacronyx chloris
- Sharpe's Longclaw, Hemimacronyx sharpei
- Abyssinian Longclaw, Macronyx flavicollis
- Fuelleborn's Longclaw, Macronyx fuelleborni
- Cape Longclaw, Macronyx capensis
- Yellow-throated Longclaw, Macronyx croceus
- Pangani Longclaw, Macronyx aurantiigula
- Rosy-throated Longclaw, Macronyx ameliae
- Grimwood's Longclaw, Macronyx grimwoodi
- Sprague's Pipit, Anthus spragueii
- Yellowish Pipit, Anthus lutescens
- Puna Pipit, Anthus brevirostris
- Short-billed Pipit, Anthus furcatus
- Peruvian Pipit, Anthus peruvianus
- Pampas Pipit, Anthus chacoensis
- Paramo Pipit, Anthus bogotensis
- Hellmayr's Pipit, Anthus hellmayri
- Correndera Pipit, Anthus correndera
- Ochre-breasted Pipit, Anthus nattereri
- Pechora Pipit, Anthus gustavi
- Alpine Pipit, Anthus gutturalis
- Madanga, Anthus "ruficollis"
- Nilgiri Pipit, Anthus nilghiriensis
- Tree Pipit, Anthus trivialis
- Olive-backed Pipit, Anthus hodgsoni
- Rosy Pipit, Anthus roseatus
- Red-throated Pipit, Anthus cervinus
- American Pipit / Buff-bellied Pipit, Anthus rubescens
- Meadow Pipit, Anthus pratensis
- Water Pipit, Anthus spinoletta
- Eurasian Rock Pipit, Anthus petrosus
Sibley and Monroe's Fringillidae
Sibley and Monroe's (1990) Fringillidae included all of the remaining birds, over 1000 of them. Most authors use a finely-grained family structure for these species that roughly corresponds to Sibley and Monroe's tribes. The TIF taxonomy divides Sibley and Monroe's Fringillidae into 11 families.
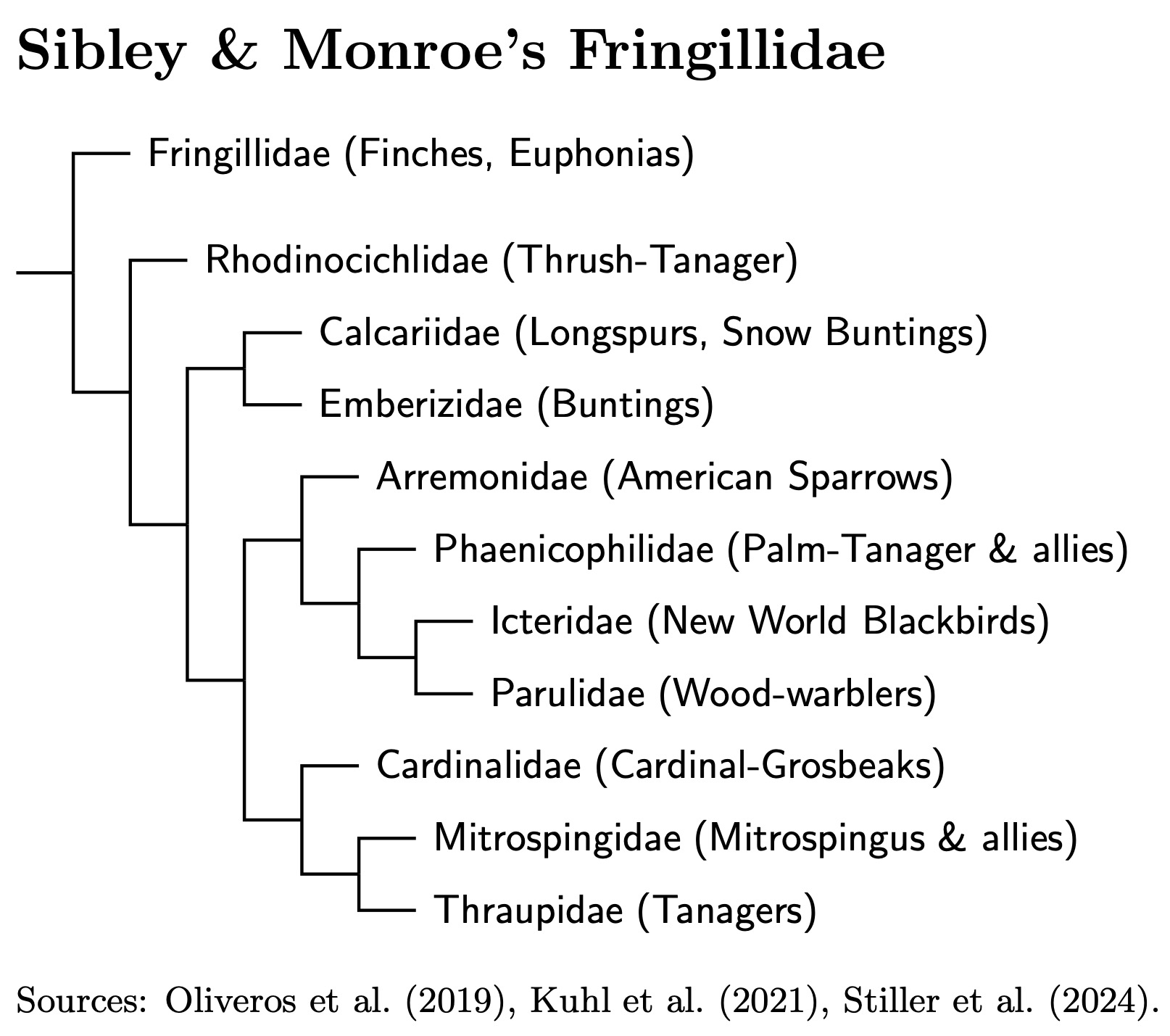 |
| Sibley and Monroe's Fringillidae: TiF Family Tree |
|---|
Fringillidae: Finches, Euphonias Leach, 1819
60 genera, 230 species HBW-15
Not long ago, the chlorophonias and euphonias were considered tanagers. Many guidebooks still list them as such, but it is not so. The AOU recognized them as finches in the 44th checklist supplement (2003), placing them in the subfamily Euphoniinae. Accordingly, they are placed in Fringillidae as a subfamily (Groth, 1998; Klicka et al., 2000; Yuri and Mindell, 2002; Zuccon et al., 2012). The Hawaiian Honeycreepers were once considered a separate family (Drepanididae). They are now thought to form a clade buried inside Carduelinae (Yuri and Mindell, 2002; Arnaiz-Villena et al., 2007b; Lerner et al., 2011; Zuccon et al., 2012). They are treated here as a tribe within Carduelinae.
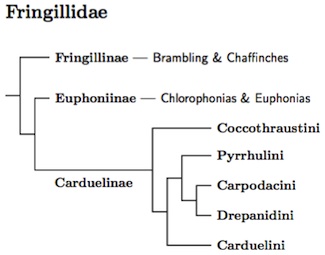 |
| Click for Fringillidae genus tree |
|---|
The arrangement of the finches is based on several sources. I originally relied on Groth (1998), Klicka et al. 2007, and Arnaiz-Villena et al. (1998, 1999, 2001, 2007a, b, 2008). Their studies have included a substantial number of finch species. However, some monotypic genera had been left out (e.g., Callacanthis, Chaunoproctus, Kozlowia, Neospiza). These have all been included in the analysis by Zuccon et al. (2012).
The tribes within the Carduelinae are now based on Lerner et al. (2011) and Zuccon et al. (2012). Except for the basal position of the Coccothraustini, there had previously been a lack of consensus on the relationships between the Cardueline tribes. See Yuri and Mindell (2002), Arnaiz-Villena et al. (2007a), Nguembock et al. (2009a), and Töpfer et al. (2011). That has changed with the publication of Lerner et al. (2011) and Zuccon et al. (2012). Although the taxon sampling is different, the two papers are completely consistent at the generic level except for the placement of the Pyrrhulini. Zuccon et al. put the Pyrhullini sister to the Carduelini while Lerner et al. place it sister to the Carpodacini/Drepanidini clade. Note that “European Serin” on Lerner et al.'s tree is a typo as noted in the supplementary material. It actually refers to the White-bellied Canary.
The combination of a large number of genes and attention to the problem of discerning the true species tree in spite of incongruent gene trees has led me to use the overall structure from Lerner et al. (2011). They use a large superset of the genes used by Zuccon et al., but only sample half as many taxa. The TiF tree of genera is 100% consistent with Lerner et al., and except for the placement of Pyrrhulini, is also consistent with Zuccon et al. It's nice to see so much consensus.
The Fringillidae start with a basal Palearctic group, Fringillinae. Fringillinae is comprised of three species, one of which has spread across the northern Palearctic. The remaining Fringillidae fall into two sister clades, Euphoniinae and Carduelinae. The common ancestor of the Euphoniinae/Carduelinae clade was likely an Old World species. Something must connect it with the Neotropical Euphoniinae, but that something is missing without a trace. Being geographically contiguous and more similar in appearance, the position of the Carduelinae is easy to understand. Euphoniinae is a mystery that unscores the importance of extinction in creating the bird distribution we currently see.
Fringillinae: Chaffinches Leach, 1819
The split of the Blue Chaffinch, Fringilla teydea, from Common Chaffinch, Fringilla coelebs, was based on on Suárez et al. (2009). The further split of Gran Canaria Blue Chaffinch, Fringilla polatzeki, from Blue Chaffinch, Fringilla teydea, is based on Suárez et al. (2009) and the analysis by Sangster et al. (2016b).
- Brambling, Fringilla montifringilla
- Common Chaffinch, Fringilla coelebs
- Blue Chaffinch, Fringilla teydea
- Gran Canaria Blue Chaffinch, Fringilla polatzeki
Euphoniinae: Euphonias, Chlorophonias Tschudi, 1846
Zuccon et al. (2012) found evidence that the Antillean Euphonia is more closely related to the chlorophonias than to most euphonias. The two other blue-headed species are usually considered closely related to musica (sometimes treated as one species under the name Blue-hooded Euphonia), so all three have been placed in the genus Cyanophonia (Bonaparte 1851, type musica).
Due mainly to plumage differences between the subspecies, the Antillean Euphonia, Cyanophonia musica has been split into Hispaniolan Euphonia, Cyanophonia musica, Puerto Rican Euphonia, Cyanophonia sclateri, and Lesser Antillean Euphonia, Cyanophonia flavifrons, following the 64th AOS Supplement.
- Golden-rumped Euphonia, Cyanophonia cyanocephala
- Elegant Euphonia, Cyanophonia elegantissima
- Hispaniolan Euphonia, Cyanophonia musica
- Puerto Rican Euphonia, Cyanophonia sclateri
- Lesser Antillean Euphonia, Cyanophonia flavifrons
- Blue-naped Chlorophonia, Chlorophonia cyanea
- Chestnut-breasted Chlorophonia, Chlorophonia pyrrhophrys
- Yellow-collared Chlorophonia, Chlorophonia flavirostris
- Blue-crowned Chlorophonia, Chlorophonia occipitalis
- Golden-browed Chlorophonia, Chlorophonia callophrys
- Jamaican Euphonia, Euphonia jamaica
- Plumbeous Euphonia, Euphonia plumbea
- Scrub Euphonia, Euphonia affinis
- Yellow-crowned Euphonia, Euphonia luteicapilla
- Purple-throated Euphonia, Euphonia chlorotica
- Trinidad Euphonia, Euphonia trinitatis
- Velvet-fronted Euphonia, Euphonia concinna
- Orange-crowned Euphonia, Euphonia saturata
- Finsch's Euphonia, Euphonia finschi
- Violaceous Euphonia, Euphonia violacea
- Thick-billed Euphonia, Euphonia laniirostris
- Yellow-throated Euphonia, Euphonia hirundinacea
- Green-throated Euphonia / Green-chinned Euphonia, Euphonia chalybea
- Fulvous-vented Euphonia, Euphonia fulvicrissa
- Spot-crowned Euphonia, Euphonia imitans
- Olive-backed Euphonia, Euphonia gouldi
- Golden-bellied Euphonia / White-lored Euphonia, Euphonia chrysopasta
- Bronze-green Euphonia, Euphonia mesochrysa
- White-vented Euphonia, Euphonia minuta
- Tawny-capped Euphonia, Euphonia anneae
- Orange-bellied Euphonia, Euphonia xanthogaster
- Rufous-bellied Euphonia, Euphonia rufiventris
- Chestnut-bellied Euphonia, Euphonia pectoralis
- Golden-sided Euphonia, Euphonia cayennensis
Carduelinae Vigors, 1825
As mentioned above, how the tribes with the Carduelinae relate has been somewhat contentious. There is considerable agreement that the holarctic Coccothraustini are basal. The results of Nguembock et al. (2009a) suggested that Carpodacus and Pyrrhula were fairly closely related to each other, as did Töpfer et al. (2011). Neither had much to say about their relation to the Hawaiian Honeycreepers (Drepanidini). Zuccon et al. (2012) were unable to clearly resolve the position of the honeycreepers (see Fig. 2), but Lerner et al. (2011) place them sister to the rosefinches (Carpodacini). The two together are then sister to the bullfinches and many arid-zone finches (Pyrrhulini). Lerner et al. place the whole lot of them are sister to Carduelini, which includes the American red finches (previously separated as a tribe). As mentioned before, Zuccon et al. disagree, placing Pyrrhulini sister to Carduelini.
Coccothraustini: Grosbeak Finches Swainson, 1831
Zuccon et al. (2012) found that Eophona was embedded within Coccothraustes. A reasonable solution to this problem is to restore the two American species to Hesperiphona and leave the Asian species in a reduced Eophona.
- Black-and-yellow Grosbeak, Mycerobas icterioides
- Collared Grosbeak, Mycerobas affinis
- Spot-winged Grosbeak, Mycerobas melanozanthos
- White-winged Grosbeak, Mycerobas carnipes
- Evening Grosbeak, Hesperiphona vespertina
- Hooded Grosbeak, Hesperiphona abeillei
- Hawfinch, Coccothraustes coccothraustes
- Chinese Grosbeak, Eophona migratoria
- Japanese Grosbeak, Eophona personata
Pyrrhulini: Bullfinches and Arid-zone FinchesVigors, 1825
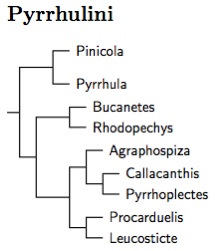 |
| Click for Pyrrhulini tree |
|---|
Pyrrhulini consists of 3 clades. The first consists of the Pyrrhula bullfinches and Pine Grosbeak (Pinicola). The second and third are sister, and are the arid-zone finches indentified by Arnaiz-Villena et al. (2008). The second clade includes Bucanetes and Rhodopechys. The third consists of the mountain-finches and rosy-finches (Leucosticte), together with several monotypic genera: Procarduelis (Dark-breasted Rosefinch), Agraphospiza (Blanford's Rosefinch), Callacanthis (Spectacled Finch), and Pyrrhoplectes (Golden-naped Finch). Both Procarduelis and Agraphospiza are usually included in Carpodacus.
The bullfinches are arranged based on Töpfer et al. (2011). Kirwan and Gregory (2005) established the monotypic genus Eremopsaltria for the Mongolian Finch, separating it from Bucanetes. However, they acted under the misapprehension that the Mongolian Finch is more likely closer to the Common Rosefinch than to the Trumpeter Finch. Arnaiz-Villena et al. (2008) found a rather different result, with the two Bucanetes as sister taxa, and the rosefinch placed rather distantly from them. Zuccon et al. (2012) confirm this.
Arnaiz-Villena et al. (2008) found that the Dark-breasted Rosefinch, formerly Carpodacus nipalensis belongs in the same clade close to Leucosticte. Rather than folding it in Leucosticte, I restored the genus name Procarduelis (Blyth 1843). I had previously noted that Blanford's Rosefinch is thought to be close to nipalensis. Indeed, Zuccon et al. found it in the other portion of this clade. It is sufficently distant from its sister group (Callacanthis and Pyrrhoplectes) to deserve a separate genus. Zuccon et al. (2012) established the name Agraphospiza for it.
I follow AOU in listing three North American rosy-finches. Some have even suggested further splitting the Gray-crowned Rosy-Finch. They have also been all been lumped into one species in the past. Drovetski et al. (2009) find little genetic difference between the three, suggesting that they may be lumped again.
- Pine Grosbeak, Pinicola enucleator
- Brown Bullfinch, Pyrrhula nipalensis
- White-cheeked Bullfinch, Pyrrhula leucogenis
- Eurasian Bullfinch, Pyrrhula pyrrhula
- Azores Bullfinch, Pyrrhula murina
- Orange Bullfinch, Pyrrhula aurantiaca
- Red-headed Bullfinch, Pyrrhula erythrocephala
- Gray-headed Bullfinch, Pyrrhula erythaca
- Trumpeter Finch, Bucanetes githagineus
- Mongolian Finch, Bucanetes mongolicus
- Eurasian Crimson-winged Finch, Rhodopechys sanguineus
- African Crimson-winged Finch, Rhodopechys alienus
- Blanford's Rosefinch, Agraphospiza rubescens
- Spectacled Finch, Callacanthis burtoni
- Golden-naped Finch, Pyrrhoplectes epauletta
- Dark-breasted Rosefinch, Procarduelis nipalensis
- Plain Mountain-Finch, Leucosticte nemoricola
- Brandt's Mountain-Finch, Leucosticte brandti
- Asian Rosy-Finch, Leucosticte arctoa
- Gray-crowned Rosy-Finch, Leucosticte tephrocotis
- Black Rosy-Finch, Leucosticte atrata
- Brown-capped Rosy-Finch, Leucosticte australis
Carpodacini: Rosefinches Bonaparte, 1853
The arrangement of the rosefinches is now based on Päckert et al. (2020b), which improves on the analyses of Tietze et al. (2013) and Zuccon et al. (2012). Now that we had a decent understanding of how the rosefinches relate, and how old the taxa are, we can establish generic limits in a reasonable way, balancing age and coherence.
There is a deep division in the basal clade, which consists of the Common Rosefinch (Erythrina) and the Scarlet Finch (Haematospiza).
The extinct Bonin Grosbeak (aka Chaunoproctus) is basal in the main clade, and is sister to the remaining two dozen rosefinches. These split into two clades.
The first ranges from Blyth's Rosefinch to Taiwan Rosefinch. The Taiwan Rosefinch, Carpodacus formosanus, has been split from Vinaceous Rosefinch, Carpodacus vinaceus, based on Wu et al. (2011).
The second clade includes synoicus group, the former Kozlowia (C. roborowskii), the Rubicilla clade (Streaked and Great Rosefinches), the former Uragus (C. sibiricus), Pyrrhospiza (C. puniceus), and Pinicola subhimachala (aka Propyrrhula subhimachala). It ends with a group including the Carpodacus type species Pallas's Rosefinch, Carpodacus roseus (see Banks and Browning 1995), through C. dubius.
Tietze et al. (2013) found substantial genetic distance between the Sinai Rosefinch in the strict sense C. synoicus synoicus, and the Pale Rosefinch group (subspecies beicki, salimalii, and presumably stoliczkae). These have been suspected of being different species, and I have now split them.
I moved Sillem's Mountain-Finch (rediscovered in June 2012) from Leucosticte (Pyrrhulini) to Carpodacus based on a photo of what is believed to be the female. When Roselaar (1992) named Sillem's Rosefinch, he put it in Leucosticte. At the time, the female plumage was unknown. The other Leucosticte have female plumages that are only slightly different from the male, usually just duller. As Roselaar pointed out, if it has a distinctive female plumage, it would more likely related to Kozlowia (now part of Carpodacus) than to Leucosticte. More recently, Muzika (2014) has made the same point. Yann Muzika's photo of the probable female Sillem's shows a bird that is quite different from the adult male. See Kazmierczak and Muzika (2012) for an account of this rediscovery and Muzika (2014) for further observations.
A DNA analysis by Sangster et al. (2016a) has confirmed that Sillem's Mountain-Finch is a Carpodacus rosefinch, and as expected, is sister to Tibetan Rosefinch, Carpodacus roborowskii.
Tietze et al. (2013) also found a surprisingly small genetic distance between the Caucasian and Spotted Great-Rosefinches (rubicilla and severtzovi) and recommended lumping them. Although I have followed that advice, further study is in order. Rasmussen and Anderton (2005) recommended splitting them based on differences in plumage, habitat, and voice.
The genus Carpodacus has been divided into the following genera:
- Common Rosefinch, Carpodacus erythrinus, is transferred to Erythrina (Brehm 1828, monotypic)
- Scarlet Finch, Carpodacus sipahi, is transferred to Haematospiza (Blyth 1845, monotypic)
- Bonin Grosbeak, Carpodacus ferreorostris, is transferred to Chaunoproctus (Bonaparte 1850, monotypic)
- Sinai Rosefinch, Carpodacus synoicus, and Pale Rosefinch, Carpodacus stoliczkae need a genus name. For now, I'll list them as "Carpodacus"
- Sillem's Mountain-Finch, Carpodacus sillemi, and Tibetan Rosefinch, Carpodacus roborowskii, are transferred to Kozlowia (Bianchi 1907, type roborowskii)
- Red-fronted Rosefinch, Carpodacus punicea, and Crimson-browed Finch, Carpodacus subhimachala, are transferred to Propyrrhula (Hodgson 1844, type subhimachala)
- Long-tailed Rosefinch, Carpodacus sibiricus, is transferred to Uragus (Keyserling and Blasius 1840, monotypic)
- Further, Long-tailed Rosefinch, Uragus sibiricus is split into Siberian Long-tailed Rosefinch, Uragus sibiricus, including sanguinolentus and ussuriensis, and Chinese Long-tailed Rosefinch, Uragus lepidus, including henrici, based on Liu et al. (2020). They suggest synonymizing ussuriensis with sanguinolentus, although IOC has not followed that suggestion.
- Pallas's Rosefinch, Carpodacus roseus and the three following species remain in Carpodacus (Kaup 1829, type roseus)
- Streaked Rosefinch, Carpodacus rubicilloides, and Great Rosefinch, Carpodacus rubicilla, are transferred to Rubicilla (Bonaparte and Schlegel 1850 type rubicilla)
- Blyth's Rosefinch, Carpodacus grandis, through Taiwan Rosefinch, Carpodacus formosanus (11 species) are transferred to Propasser (Hodgson 1844 type rodopeplus)
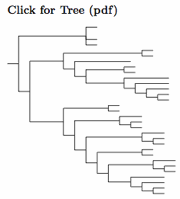 |
| Click for Carpodacini tree |
|---|
- Common Rosefinch, Erythrina erythrina
- Scarlet Finch, Haematospiza sipahi
- †Bonin Grosbeak, Chaunoproctus ferreorostris
- Sinai Rosefinch, "Carpodacus" synoicus
- Pale Rosefinch, "Carpodacus" stoliczkae
- Sillem's Mountain-Finch, Kozlowia sillemi
- Tibetan Rosefinch, Kozlowia roborowskii
- Red-fronted Rosefinch, Propyrrhula punicea
- Crimson-browed Finch, Propyrrhula subhimachala
- Siberian Long-tailed Rosefinch, Uragus sibiricus
- Chinese Long-tailed Rosefinch, Uragus lepidus
- Pallas's Rosefinch, Carpodacus roseus
- Three-banded Rosefinch, Carpodacus trifasciatus
- Himalayan White-browed Rosefinch, Carpodacus thura
- Chinese White-browed Rosefinch, Carpodacus dubius
- Streaked Rosefinch, Rubicilla rubicilloides
- Great Rosefinch, Rubicilla rubicilla
- Blyth's Rosefinch, Propasser grandis
- Red-mantled Rosefinch, Propasser rhodochlamys
- Pink-rumped Rosefinch, Propasser waltoni
- Himalayan Beautiful-Rosefinch, Propasser pulcherrimus
- Chinese Beautiful-Rosefinch, Propasser davidianus
- Dark-rumped Rosefinch, Propasser edwardsii
- Pink-browed Rosefinch, Propasser rodochroa
- Sharpe's Rosefinch, Propasser verreauxii
- Spot-winged Rosefinch, Propasser rodopeplus
- Vinaceous Rosefinch, Propasser vinaceus
- Taiwan Rosefinch, Propasser formosanus
Drepanidini Cabanis, 1847
The results in both Lerner et al. (2011) and Zuccon et al. (2012) support the Drepanidini as sister to Carpodacini (not just the Common Rosefinch, as might be inferred from the press coverage). Interestingly, the extinct Bonin Grosbeak is a relatively basal member of the Carpodacini. Perhaps it too is a relic of the spread of the ancestral Carpodacini/Drepanidini into the Pacific.
The order here is based on a combination of the DNA study of Lerner et al. (2011) and the osteological study of James (2004). Fleischer et al. (2001), Pratt (2001), Arnaiz-Villena et al. (2007b), and Reding et al. (2009) have also been consulted. It fits nicely with morphology (look at bill shapes in each clade) and subfossil taxa can be easily accomodated using James (2004). This is pretty similar to the previous version, but tweaked based on Lerner et al. (2011), which greatly decreases the conjecture needed, and increases my confidence in it.
The genera Palmeria, Himatione, and Vestiaria have been merged into Drepanis. The genetic distance between them seems to be small (Lerner et al., 2011) and there is evidence of hybridization between Vestiaria and Himatione (Knowlton et al., 2014). Olson (2012b) also argues that this is a reasonable treatment. Although I think the genera I use for the Hawaiian Honeycreepers are oversplit, I consider the AOU genera even more oversplit. I haven't gone further on the lumping because data on the extinct species is too limited.
Although Pratt (2009) has established the genus Manucerthia for the Hawaiian Creeper, Loxops mana, I have not adopted it. Lerner et al. (2011) make clear that the Hawaiian Creeper is sister to the other Loxops. Moreover, they shared a common ancestor about 2 million years ago and do not seem so different as to justify introducing an extra genus into a tribe that already has too many genera.
I have kept the genus Akialoa merged with Hemignathus as Akialoa appears to be paraphyletic (see the trees in James, 2004). However, portions of Hemignathus have been split off as Chlorodrepanis and Viridonia. This has allowed restoration of the name Hemignathus wilsoni for the Akiapolaau (Hemignathus munroi). When Chlorodrepanis virens is subsumed in Hemignathus, the name wilsoni belongs to a subspecies of virens. The situation with the Kauai Amakihi, Chlorodrepanis stejnegeri is similar. In Hemignathus, the name stejnegeri belongs to a subspecies of the Greater Akialoa, Hemignathus ellisianus stejnegeri, and the Kauai Amakihi uses the substitute name H. kauaiensis.
I follow the AOU decisions (56th supplement) to split the Apapane, Akepa, Greater Akialoa, and Nukupuu. Thus:
- Apapane, Drepanis (Himatione) sanguinea is split into
- Laysan Honeycreeper, Drepanis fraithii (extinct)
- Apapane, Drepanis sanguinea
- Akepa / Hawaii Akepa, Loxops coccineus is split into
- Oahu Akepa, Loxops wolstenholmei (probably extinct)
- Maui Akepa, Loxops ochraceus (probably extinct)
- Hawaii Akepa, Loxops coccineus
- Nukupuu, Hemignathus lucidus, is split into
- Kauai Nukupuu, Hemignathus hanapepe (probably extinct)
- Oahu Nukupuu, Hemignathus lucidus (probably extinct)
- Maui Nukupuu, Hemignathus affinis (possibly extinct)
- Greater Akialoa, Hemignathus ellisianus, is split into
- Kauai Akialoa, Hemignathus stejnegeri (extinct)
- Oahu Akialoa, Hemignathus ellisianus (extinct)
- Maui-nui Akialoa, Hemignathus lanaiensis (extinct)
This changes clade sizes and I have also reordered the Hawaiian Honeycreepers as a result.
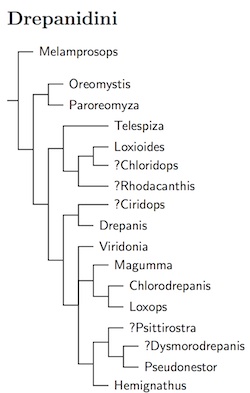 |
| Click for Drepanidini species tree |
|---|
- Poo-uli, Melamprosops phaeosoma
- Akikiki / Kaui Creeper, Oreomystis bairdi
- Oahu Alauahio / Oahu Creeper, Paroreomyza maculata
- Kakawahie / Molokai Creeper, Paroreomyza flammea
- Maui Alauahio / Maui Creeper, Paroreomyza montana
- Laysan Finch, Telespiza cantans
- Nihoa Finch, Telespiza ultima
- Palila, Loxioides bailleui
- Kona Grosbeak, Chloridops kona
- Lesser Koa-Finch, Rhodacanthis flaviceps
- Greater Koa-Finch, Rhodacanthis palmeri
- Ula-ai-hawane, Ciridops anna
- Akohekohe / Crested Honeycreeper, Drepanis dolei
- Laysan Honeycreeper, Drepanis fraithii
- Apapane, Drepanis sanguinea
- Iiwi, Drepanis coccinea
- Black Mamo, Drepanis funerea
- Hawaii Mamo, Drepanis pacifica
- Greater Amakihi, Viridonia sagittirostris
- Anianiau, Magumma parva
- Hawaii Amakihi, Chlorodrepanis virens
- Kauai Amakihi, Chlorodrepanis stejnegeri
- Oahu Amakihi, Chlorodrepanis flava
- Hawaii Creeper, Loxops mana
- Akekee / Kauai Akepa, Loxops caeruleirostris
- Oahu Akepa, Loxops wolstenholmei
- Maui Akepa, Loxops ochraceus
- Hawaii Akepa, Loxops coccineus
- Ou, Psittirostra psittacea
- Lanai Hookbill, Dysmorodrepanis munroi
- Maui Parrotbill / Kiwikiu, Pseudonestor xanthophrys
- Lesser Akialoa / Hawaii Akialoa, Hemignathus obscurus
- Kauai Akialoa, Hemignathus stejnegeri
- Oahu Akialoa, Hemignathus ellisianus
- Maui-nui Akialoa, Hemignathus lanaiensis
- Akiapolaau, Hemignathus wilsoni
- Kauai Nukupuu, Hemignathus hanapepe
- Oahu Nukupuu, Hemignathus lucidus
- Maui Nukupuu, Hemignathus affinis
Carduelini: Canaries, Siskins and allies Vigors, 1825
This brings us to the Carduelini. It has become apparent that the North American Carpodacus finches are not that closely related to the true Carpodacus finches. Previously, the TiF list used the genus Burrica, also used as a subgenus by AOU (2nd-5th AOU checklists). However, the oldest available name seems to be Erythrospiza (Bonaparte 1830, type purpurea—compare Haemorhous Swainson, 1837, type purpureus or Burrica Ridgway, 1887, type mexicana). Erythrospiza appears to be sister to the rest of the Carduelini, and rather distantly related. Perhaps it would even make sense to treat it as a separate tribe (as done previously in the TiF list).
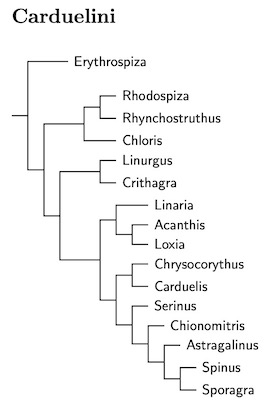 |
| Click for Carduelini tree |
|---|
I follow the recent paper by Zuccon et al. (2012) to arrange the various Cardueline genera. Lerner et al. (2011) gives essentially identical results for the taxa they included. Previous studies did a decent job at the generic level, and sometimes for groups of genera, but were unable to get strong support for any method of arranging them. Thus the papers by Arnaiz-Villena et al. (1998, 1999, 2001, 2007a, b, 2008) and Zamora et al. (2006) have been used in ordering the species and determing the exact generic boundaries. Unfortunately, the papers by Arnaiz-Villena and colleagues have been completely useless for determining deeper relations. One wonders whey they continue to reuse the same data rather than collecting more. Nguembock et al. (2009a) has also been helpful at higher levels, but I rely on Lerner et al. and Zuccon et al. for the overall structure.
The various studies of the finches have made it clear that the genera Carduelis and Serinus both required substantial surgery. The alternative for Carduelis would be to put almost all of the Carduelini into one genus! For Serinus, the alternative is less drastic, mainly because Carduelis has priority. Neither of these outcomes is particuarly desirable, so both Serinus and Carduelis have been divided into several pieces each.
As constituted here, Carduelini contains 4 major clades. As mentioned above, the first clade includes only a single genus: Erythrospiza — the American purple finches. The genus has been changed from Haemorhous (Swainson 1837) to Erythrospiza (Bonaparte 1830) type purpurea as the latter has priority.
Greenfinches and Golden-winged Grosbeaks: The second clade includes the Chloris greenfinches (sometimes included in Carduelis), the Desert Finch (Rhodospiza), and the the Golden-winged Grosbeaks (Rhynchostruthus). Kirwan and Grieve (2007) argue that Rhynchostruthus includes three species.
Siskins and allies: The third Carduelini clade is exclusively Afrotropical. The Oriole Finch (Linurgus) is sister to the rest. These closely related birds are variously called canaries, seedeaters, serins, and siskins and are all placed in the genus Crithagra. The species-level phylogeny still needs some work, as might be guessed from the question marks, polytomies, and blue ink.
I recognize several subgenera. Besides Crithagra, there are Dendrospiza, Neospiza, Pseudochloroptila, and Ochrospiza. All are marked on the species tree.
Subgenus Crithagra includes most of the larger African canaries and allies.
The Sao Tome Grosbeak has usually been included in Neospiza. Stervander (2010) found that the Principe Seedeater is it's closest relative and both species together form subgenus Neospiza. In fact, Stervander found that the Sao Tome subspecies of the seedeater (thomensis) appeared more closely related to the Sao Tome Grosbeak than to the seedeater. The two birds are quite distinct, and it seems unreasonable to put them in one species. Perhaps the genetic results are a sign of introgression.
It's not entirely clear whether subgenus Pseudochloroptila belongs in subgenus Ochrospiza, so they are kept separate. Both the Yemen Serin, Crithagra menachensis, and Ankober Serin, Crithagra ankoberensis, remain somewhat mysterious. These have be considered conspecific by some, or in separate genera by others. I have little confidence in their placement here (hence the blue color on the tree), but it seems at least as reasonable as any other. This clade is expected to get a revision when more information becomes available.
Linnets: The fourth Carduelini clade is more cosmopolitan. The basal piece includes the Palearctic Linnets (Linaria, formerly Carduelis), which are sister to the holarctic Redpolls (now Acanthis rather than Carduelis) and crossbills (Loxia). There is some uncertainty about the linnets. The mitochondrial dna consistently puts them near the siskins and American goldfinches rather than near the redpolls and crossbills. The nuclear dna does the opposite. Since they seem more akin to the redpolls and crossbills, and since the combined analysis puts them there, I have followed suit.
Redpolls: The redpolls themselves are an interesting case. It's not clear how many redpoll species there are. There is evidence that at least two are good biological species (e.g., Knox, 1988). In fact, Knox et al. (2001) support separation of Lesser Redpoll, Acanthis cabaret. However, genetic studies have failed to find any differences between Lesser and Common Redpolls (Ottvall et al., 2002). Worse, Marthinsen et al. (2008) found little genetic difference between any of the redpolls! There may be only one redpoll species. Indeed, Mason and Taylor's detailed study using SNP's (2015) found little genetic differentiation among the redpolls. At this point the balance of the evidence is that there is only one species involved. As a result, I've lumped them all as a single species, Redpoll, Acanthis flammea.
Crossbills: A number of genetic studies have been done on the crossbills, and it's time the crossbills got a new taxonomy. Since Groth (1988, 1993) ornithologists have tried to use call notes to separate crossbill species. DNA has made clear that call notes are sometimes relevant, but are often not. This may be partly due to the fact that crossbill call notes are learned (Keenan and Benkman, 2008; Martin et al., 2024).
Fortunately, we have DNA to enlighten us. The basic genetic structure of Loxia had already appeared in Parchman, Benkman, and Mezquida (2007), but no one realized it. This may be because the paper didn't deal with all of the crossbills that were then recognized as species. It lacked both Scottish and Parrot Crossbills. It also didn't it go into detail about the crossbill flight calls or the relation between crossbills and pines, other than on the island of Hispaniola. It did successfully argue that the Hispaniolan Crossbill, Loxia megaplaga, is a separate species. However, there is more we can learn from this paper.
Let's see what Figure 4 of Parchman et al. (2007) can tell us. It starts with a basal split in the crossbills that happened about 1.5 mya (I've scaled their calibration numbers based on Fig. 4). That split separated the plain-winged crossbills and the barred-winged crossbills. The barred-winged clade splits at about 0.9 mya into Old and New World clades, and the New World clade splits again around 0.7 mya into a northern group and one now confined to Hispaniola.
Parchman et al. argued for species status for the Hispaniolan Crossbill, Loxia megaplaga, which is well-separated from the other North American crossbills, and closely tied to the Hispaniolan Pine, Pinus occidentalis. However, the earlier split, into Old and New World populations is old enough to give them species status too. That split occurred nearly a million years ago. The two taxa are also well-separated geographically, suggesting little gene flow. That gives us the Two-barred Crossbill, Loxia bifasciata in Eurasia (mostly breeding in Russia) and the White-winged Crossbill, Loxia leucoptera in North America. This gives all three recognized subspecies of L. leucoptera species status.
Continuing with Parchman et al. (2007), they also found a split in the plain-winged crossbills about that occurred slightly later than that in the barred-winged clade, roughly 0.85 mya. That suggests the New and Old World clades are also separate species, the Common Crossbill, Loxia curvirostra of Eurasia and North Africa, and the Red Crossbill, Loxia minor of North and Middle America. The geographic separation is not as strong for plain-winged crossbills, but we'll see later that there's genetic evidence of a lack of gene-flow between them.
Version 14.2 of the IOC list includes 22 subspecies in the plain-winged group, 13 in the Old World clade, and 9 in the New World clade. The IOC gives three of them species status, but the data leads me to a different conclusion. To start, we turn to the phylogenetic tree Laurent Raty, constructed and posted on BirdForum in 2017. It mostly includes plain-winged crossbills. The arrangement of the few barred-winged crossbills is only weakly supported. He has helpfully colored the Red Crossbills using orange for the New World taxa and green for the Old World taxa. You'll notice that none end up in the other group, supporting the idea that there is little gene flow between them.
Raty's tree also lets us address the status of the Scottish and Parrot crossbills. Before continuing, we should recall the biological species concept. A species consists of group(s) of interbreeding populations that are reproductively isolated from other populations. That isolation can occur in different ways, it could be geographic isolation, or it could be that the species cannot produce fertile offspring, as is the case when Mallards interbreed with Muscovy Ducks. There are sometimes offspring, but they are infertile.
Summers et al. (2007) argued that the three crossbill subspecies in Scotland, curvirostra, scotica, and pytyopsittacus, are separate species based on positive assortative mating. I found this unconvincing. Their sample sizes are not large enough to show that gene flow is very small. Indeed, if I understand correctly, gene flow between the taxa could be over 4%. I'm not an expert on these matters, but from what I've read, that's more that enough to maintain the ability to interbreed. In other words, the populations curvirostra, scotica, and pytyopsittacus are in the same biological species. In that case, the lack of strong geographical separation suggests they may be best thought of as morphs, not subspecies. But there's also the assortative mating, so I'm just not sure.
Since assortative mating didn't help, we return to the DNA. Potential species show up in phylogenetic trees as clades, subtrees of the phylogenetic tree, or at least nearly so. If two types of crossbills have not interbred for a long time, they should each be in their own subtree. This is called reciprocal monophyly. If one species has recently separated from a parent species, it should have its own subtree, but the parent species may consist of two or more subtrees, a phenomenon known as incomplete lineage sorting. It takes time for the still interbreeding parts of the parent species to become each others closest relatives again.
But we don't see anything resembling a subtree for either the Scottish Crossbill, L. scotica, or the Parrot Crossbill, L. pytyopsittacus in the Raty tree. They are scattered throughout the Old World portion of the tree. To drive home the point, focus on pytyopsittacus. One is near a japonica, one is sister to a luzonensis, others group with a himalayensis, the rest are scattered about the tree. They don't group together, and neither do the Scottish Crossbills. They are not species.
As if that's not enough, Parchman et al. (2018) analyzed over 18,000 single nucleotide polymorphisms (sites that differed across the crossbill specimens), sampled from Europe and N. Africa. The results are illustrated in their Figure 3. Although it contains data from across Europe and North Africa, it focuses on the subspecies of Spain and Morocco. Again scotica and pytyopsittacus did not form clades. This means that two independent genetic analyses on different data sets fail to support the hypothesis that the Scottish and Parrot crossbills are species. There's just no strong evidence in favor them being biological species.
Before leaving Parchman et al. (2018), we should note that two of the species sampled by Parchman et al. (2018), show up as clades. These are Moroccan subspecies poliogyna, sampled from the Atlas Mountains, and balearica, sampled from Mallorca. The subspecies hispana, sampled from Málaga is in 3 pieces. The crossbills from Morocco and the Baleric Islands may deserve more attention. It's possible that hispana may also be in the process of speciation.
Finally, we turn our attention to the North American plain-winged crossbills, L. minor.* Benkman et al. (2009) made a case for considering the call type 9 crossbills of southern Idaho to be a separate species, Cassia Crossbill, Loxia sinesciuris. The AOS finally accepted it as a species in 2017, following the appearance of Parchman (2016). The proposal, and comments about are available on the AOS website. See proposal 2017-A-4.
Parchman et al. (2016) used over 18,000 crossbill SNP's to construct the phylogenetic tree in their Figure 1. The same tree also appears in AOU proposal 2017-A-4. They had data representing 37 Cassia Crossbills. They form a clade. The Cassia Crossbill and the other North American Red Crossbills are not reciprocally monophyletic, reflecting the extremely recent split of the Cassia Crossbill (probably within the last 6,000 years)
The recency of the split has made me a bit reluctant to accept it, but the data strongly suggest the Cassia Crossbill is reproductively isolated. Perhaps I'm wrong to worry about that. A 2025 paper on dog evolution (Elzinga et al., 2025) argues that even without human intervention, dogs could have evolved from wolves in as little as 8,000 years.
I also worry that the isolation seems to be partly based on soft barriers, such as song and call type, which are both learned (Sewall 2009, 2010). Nonetheless, the balance of the current evidence is strongly in favor of adding the Cassia Crossbill to the TiF list, and I have done so.
I also wonder what the chances are of getting a clade from a geographically restricted group of crossbills (or any bird species). In particular, there is a second such clade in Parchman et al. (2016). It consists of 13 crossbills from the Chiricahua mountains of SE Arizona. These are presumably L. m. stricklandi, sometimes called the Mexican Crossbill, call type 6. Are they also a separate species? Or would additional samples from Mexico overturn the result? What about the crossbills south and east of the Isthmus of Tehuantepec? That is subspecies mesamericana, call type 11? These questions await further analysis.
Keeping in mind that the Scottish Crossbill, Loxia scotica, and Parrot Crossbill, Loxia pytyopsittacus are now included in the Common Crossbill, Loxia curvirostra, we are left with the following six crossbill species:
- Common Crossbill, Loxia curvirostra, including pytyopsittacus, scotica, balearica, corsicana, poliogyna, guillemardi, altaiensis, tianschanica, himalayensis, meridionalis, japonica, and luzoniensis.
- Red Crossbill, Loxia minor, including percna, sitkensis, bendirei, benti, grinnelli, and, for now, stricklandi and mesamericana.
- Cassia Crossbill, Loxia sinesciuris (monotypic)
- Two-barred Crossbill, Loxia bifasciata (monotypic)
- White-winged Crossbill, Loxia leucoptera (monotypic)
- Hispaniolan Crossbill, Loxia megaplaga (monotypic)
Carudelis Finches: The other piece of this final clade includes the true Carduelis and Serinus finches of the Palearctic. It starts with the Mountain Serin of Indonesia and the Philippines. The Mountain Serin is sometimes considered part of Serinus, but I've had it in its own genus, Chrysocorythus, for a while. Zuccon et al. (2012) found Chrysocorythus sister to the true Carduelis finches, now reduced to the European Goldfinch and the Corsican and Citril Finches. These two genera together are sister to the rest of the clade.
The now-reduced Serinus comes next, followed by the Tibetan Serin. It's in Chionomitris rather than Serinus.
Most of the remaining Carduelini are from the Americas. There are two classification schemes in current use. The AOU's NACC puts them all together in genus Spinus. The AOU's SACC separates them into three genera: The North American goldfinches, Astragalinus; the northern siskins Spinus; and the Middle and South American siskins, Sporagra. Astragalinus splits off first, and the final division is between Spinus and Sporagra.
Several Carduelis siskins have moved to Spinus (including Lesser Antillean Siskin). In another taxonomic note, the Pine Siskin, Spinus pinus, and Black-capped Siskin, Spinus atriceps, are quite close. Some have suggested they are conspecific. Although they don't comment on it, the genetic tree in Arnaiz-Villena et al. (2008) suggests another possibility. The subspecies perplexus may actually belong to S. atriceps, as had been suggested by Banks in 1982 (see a 2008 AOU proposal).
The placement of Spinus may be a little soft. E.g., in Nguembock et al. (2009a), it appeared in different places in the combined tree (their Figure 4) and some of the individual gene trees. This is the subject of some discussion in Nguembock et al. The arrangement here is based on Zuccon et al. (2012) and Beckman and Witt (2015).
These three genera were formerly considered part of Carduelis. The AOU currently treats them all as Spinus, and rejected a proposal to make these last two generic splits, also proposed by Nguembock et al.
One has to be careful with the genus name for the South American siskins. Both Sporagra (Reichenbach 1850, type magellanica) and Pyrrhomitris (Bonaparte 1850, type cucullata) come into consideration. The publication date of Sporagra seems to be June 1, 1850. The publication date for Pyrrhomitris is not as clear. Bonaparte's “Conspectus generum avium” was published in sections beginning in mid-1850. The first part was already available in mid-June, and likely published a bit earlier, perhaps earlier than Reichenbach. However, Pyrrhomitris was not included in the section I, nor was it even included in the first part of section II (published by Oct 15, 1850). It appeared in the second part of section II (dated Nov 10, 1850 and certainly published before Feb 3, 1851). If I understand the ICZN correctly, the parts should be treated as separate publications, in which case it appeared either in the later part of 1850 or early 1851. That would give priority to Reichenbach's Sporagra. In this case there is further evidence of which was published first. Bonaparte refers to Sporagra on page 516, the page before he establishes Pyrrhomitris.
Based on Beckman and Witt (2015), the Hooded Siskin, Sporagra magellanica, has been split into Lowland Hooded Siskin, Sporagra magellanica, and Andean Hooded Siskin, Sporagra capitalis. We lack complete information on the subspecies, but I've tentatively allocated boliviana, alleni, icterica, longirostris, and magellanica to the lowland group, S. magellanica, and capitalis, paula, peruana, urubambensis, santaecrucis, hoyi, and tucumana to the Andean group, S. capitalis. The genetic distances between the capitalis group and atrata, crassirostris, siemiradzkii is razor-thin, calling their species status into question. Even uropygialis appears to be closely related to the capitalis group. Further study is needed to sort out these taxa.
- House Finch, Erythrospiza mexicana
- Purple Finch, Erythrospiza purpurea
- Cassin's Finch, Erythrospiza cassinii
- Desert Finch, Rhodospiza obsoleta
- Socotra Golden-winged Grosbeak, Rhynchostruthus socotranus
- Arabian Golden-winged Grosbeak, Rhynchostruthus percivali
- Somali Golden-winged Grosbeak, Rhynchostruthus louisae
- European Greenfinch, Chloris chloris
- Oriental Greenfinch / Gray-capped Greenfinch, Chloris sinica
- Yellow-breasted Greenfinch, Chloris spinoides
- Vietnamese Greenfinch, Chloris monguilloti
- Black-headed Greenfinch, Chloris ambigua
- Oriole Finch, Linurgus olivaceus
- Thick-billed Seedeater, Crithagra burtoni
- Protea Seedeater / Protea Canary, Crithagra leucoptera
- Kipengere Seedeater, Crithagra melanochroa
- Streaky Seedeater, Crithagra striolata
- Yellow-browed Seedeater, Crithagra whytii
- White-throated Canary, Crithagra albogularis
- Brimstone Canary, Crithagra sulphurata
- Yellow Canary, Crithagra flaviventris
- Northern Grosbeak-Canary, Crithagra donaldsoni
- Southern Grosbeak-Canary, Crithagra buchanani
- Brown-rumped Seedeater, Crithagra tristriata
- Reichard's Seedeater, Crithagra reichardi
- Black-eared Seedeater, Crithagra mennelli
- West African Seedeater, Crithagra canicapilla
- Streaky-headed Seedeater, Crithagra gularis
- Principe Seedeater, Crithagra rufobrunnea
- Sao Tome Grosbeak, Crithagra concolor
- Black-faced Canary, Crithagra capistrata
- Papyrus Canary, Crithagra koliensis
- Forest Canary, Crithagra scotops
- African Citril, Crithagra citrinelloides
- Western Citril, Crithagra frontalis
- Southern Citril, Crithagra hyposticta
- Cape Siskin, Crithagra totta
- Drakensberg Siskin, Crithagra symonsi
- Yemen Serin, Crithagra menachensis
- Ankober Serin, Crithagra ankoberensis
- White-bellied Canary, Crithagra dorsostriata
- Yellow-fronted Canary, Crithagra mozambica
- Lemon-breasted Canary, Crithagra citrinipectus
- White-rumped Seedeater, Crithagra leucopygia
- Black-throated Canary, Crithagra atrogularis
- Yellow-rumped Seedeater, Crithagra xanthopygia
- Reichenow's Seedeater, Crithagra reichenowi
- Arabian Seedeater / Arabian Serin, Crithagra rothschildi
- Yellow-throated Seedeater, Crithagra flavigula
- Salvadori's Seedeater, Crithagra xantholaema
- Twite, Linaria flavirostris
- Common Linnet, Linaria cannabina
- Yemen Linnet, Linaria yemenensis
- Warsangli Linnet, Linaria johannis
- Redpoll, Acanthis flammea
- Common Crossbill, Loxia curvirostra
- Red Crossbill, Loxia minor
- Cassia Crossbill, Loxia sinesciuris
- Two-barred Crossbill, Loxia bifasciata
- White-winged Crossbill, Loxia leucoptera
- Hispaniolan Crossbill, Loxia megaplaga
- Mountain Serin, Chrysocorythus estherae
- European Goldfinch, Carduelis carduelis
- Citril Finch, Carduelis citrinella
- Corsican Finch, Carduelis corsicana
- Island Canary / Atlantic Canary, Serinus canaria
- European Serin, Serinus serinus
- Syrian Serin, Serinus syriacus
- Red-fronted Serin, Serinus pusillus
- Ethiopian Siskin, Serinus nigriceps
- Black-headed Canary, Serinus alario
- Yellow-crowned Canary, Serinus flavivertex
- Cape Canary, Serinus canicollis
- Tibetan Serin, Chionomitris thibetana
- Lawrence's Goldfinch, Astragalinus lawrencei
- American Goldfinch, Astragalinus tristis
- Lesser Goldfinch, Astragalinus psaltria
- Antillean Siskin, Spinus dominicensis
- Eurasian Siskin, Spinus spinus
- Pine Siskin, Spinus pinus
- Black-capped Siskin, Spinus atriceps
- Black-headed Siskin, Sporagra notata
- Red Siskin, Sporagra cucullata
- Black-chinned Siskin, Sporagra barbata
- Andean Siskin, Sporagra spinescens
- Olivaceous Siskin, Sporagra olivacea
- Yellow-bellied Siskin, Sporagra xanthogastra
- Yellow-faced Siskin, Sporagra yarrellii
- Lowland Hooded Siskin, Sporagra magellanica
- Yellow-rumped Siskin, Sporagra uropygialis
- Andean Hooded Siskin, Sporagra capitalis
- Saffron Siskin, Sporagra siemiradzkii
- Thick-billed Siskin, Sporagra crassirostris
- Black Siskin, Sporagra atrata